- British Pharmacopoeia Volume III
- Formulated Preparations: Specific Monographs
Salbutamol Injection |
Beta2-adrenoceptor agonist; bronchodilator.
Salbutamol Injection is a sterile solution of Salbutamol Sulfate in Water for Injections.
The injection complies with the requirements stated under Parenteral Preparations and with the following requirements.
90.0 to 110.0% of the stated amount.
A colourless or very pale yellow solution.
A. Dilute a volume of the injection with sufficient 0.1m hydrochloric acid to produce a solution containing the equivalent of 0.004% w/v of salbutamol. The light absorption of the resulting solution, Appendix II B, in the range 230 to 350 nm exhibits a maximum only at 276 nm.
B. Carry out the method for thin-layer chromatography, Appendix III A, using silica gel G as the coating substance and a mixture of 50 volumes of ethyl acetate, 30 volumes of propan-2-ol, 16 volumes of water and 4 volumes of 13.5m ammonia as the mobile phase. Apply separately to the plate 2 µL of each of the following solutions. For solution (1) evaporate a suitable volume of the injection to dryness using a rotary evaporator, wash the residue with four 5-mL quantities of absolute ethanol, filter, evaporate the filtrate to dryness and dissolve the residue in sufficient water to produce a solution containing the equivalent of 0.1% w/v of salbutamol. Solution (2) contains 0.12% w/v of salbutamol sulfate BPCRS. After removal of the plate, allow it to dry in air until the solvent has evaporated, place it in an atmosphere saturated with diethylamine for a few minutes and spray with diazotised nitroaniline solution. The spot in the chromatogram obtained with solution (1) corresponds to that in the chromatogram obtained with solution (2).
C. Dilute a volume containing the equivalent of 0.5 mg of salbutamol to 50 mL with water, add 1 mL of 5m ammonia, 1 mL of a 3% w/v solution of 4-aminophenazone, 10 mL of a 2% w/v solution of potassium hexacyanoferrate(iii) and 10 mL of chloroform, shake and allow to separate. An orange-red colour is produced in the chloroform layer.
D. A volume containing the equivalent of 1 mg of salbutamol yields the reactions characteristic of sulfates, Appendix VI.
pH, 3.4 to 5.0, Appendix V L.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. For solution (1) dilute a volume of the injection, if necessary, with the mobile phase to produce a solution containing the equivalent of 0.005% w/v of salbutamol. For solution (2) dilute 1 volume of solution (1) to 100 volumes with the mobile phase. Solution (3) contains 0.0004% w/v of salbutamol impurity B BPCRS and 0.0005% w/v of salbutamol sulfate BPCRS in the mobile phase. For solution (1) allow the chromatography to proceed for 25 times the retention time of salbutamol (retention time of salbutamol, about 1.9 minutes).
The chromatographic procedure may be carried out using (a) a stainless steel column (15 cm × 3.9 mm) packed with spherical end-capped octylsilyl silica gel for chromatography (5 µm) with a specific surface area of 335 m2 per g, a pore size of 10 nm and a carbon loading of 11.7% (Waters Symmetry C8 is suitable), (b) as the mobile phase with a flow rate of 1 mL per minute a mixture of 22 volumes of acetonitrile and 78 volumes of a solution containing 0.287% w/v of sodium heptanesulfonate and 0.25% w/v of potassium dihydrogen orthophosphate adjusted to pH 3.65 with 2m orthophosphoric acid and (c) a detection wavelength of 220 nm.
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution factor between the two principal peaks is at least 3.0
In the chromatogram obtained with the solution (1) the area of any peak with a retention time relative to salbutamol of about 2.7 is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1%), the area of any other secondary peak is not greater than half the area of the principal peak in the chromatogram obtained with solution (2) (0.5%) and the sum of the areas of any such peaks is not greater than twice the area of the principal peak in the chromatogram obtained with solution (2) (2%).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. For solution (1) dilute a quantity of the injection solution, if necessary, with sufficient water to produce a solution containing the equivalent of 0.050% w/v of salbutamol. Solution (2) contains 0.00025% w/v of salbutamol ketone impurity BPCRS in water.
The chromatographic procedure may be carried out using (a) a stainless steel column (25 cm × 4.6 mm) packed with end-capped octylsilyl silica gel for chromatography (5 µm) (Hypersil BDS C8 is suitable) maintained at 30°, (b) as the mobile phase with a flow rate of 1 mL per minute the changing proportions of mobile phases A and B described below and (c) a detection wavelength of 276 nm.
Mobile phase A A mixture of 1.5 volumes of propan-2-ol and 98.5 volumes of 0.1m ammonium acetate adjusted to pH 4.5 with glacial acetic acid.
Mobile phase B propan-2-ol.
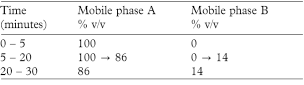
In the chromatogram obtained with solution (1) the area of any peak corresponding to salbutamol ketone is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.5%).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. For solution (1) dilute the injection with the mobile phase to produce a solution containing the equivalent of 0.0025% w/v of salbutamol. Solution (2) contains 0.003% w/v of salbutamol sulfate BPCRS in the mobile phase. Solution (3) contains 0.003% w/v of salbutamol sulfate BPCRS and 0.003% w/v of 2-tert-butylamino-1-(4-hydroxy-3-methylphenyl)ethanol sulfate BPCRS in the mobile phase.
The chromatographic procedure may be carried out using (a) a stainless steel column (20 cm × 5 mm) packed with spherical particles of silica, 5 µm in diameter, the surface of which has been modified with chemically-bonded nitrile groups (Spherisorb CN is suitable), (b) as the mobile phase with a flow rate of 2.0 mL per minute a mixture of 15 volumes of propan-2-ol, 300 volumes of 0.05m ammonium acetate and 685 volumes of water the pH of the mixture being adjusted to 4.5 with glacial acetic acid and (c) a detection wavelength of 276 nm.
The test is not valid unless the resolution factor between the two principal peaks in the chromatogram obtained with solution (3) is at least 1.5.
Calculate the content of C13H21NO3 using the declared content of C13H21NO3 in salbutamol sulfate BPCRS.
The quantity of active ingredient is stated in terms of the equivalent amount of salbutamol.