- British Pharmacopoeia Volume III
- Formulated Preparations: Specific Monographs
Salbutamol Nebuliser Solution |
Beta2-adrenoceptor agonist; bronchodilator.
Salbutamol Nebuliser Solution is a solution of Salbutamol Sulfate in Water for Injections.
The nebuliser solution complies with the requirements stated under Preparations for Inhalation and with the following requirements.
95.0 to 105.0% of the stated amount.
A. Dilute the solution being examined with sufficient 0.1m hydrochloric acid to produce a solution containing the equivalent of 0.008% w/v of salbutamol. The light absorption of the resulting solution, Appendix II B, in the range 230 nm to 350 nm exhibits a maximum only at 276 nm.
B. In the test for Related substances, the principal peak in the chromatogram obtained with solution (2) corresponds to the salbutamol peak in the chromatogram obtained with solution (3).
C. Dilute the solution being examined with sufficient water to produce a solution containing the equivalent of 0.025% w/v of salbutamol. The resulting solution yields reaction A characteristic of sulfates, Appendix VI.
pH, 3.0 to 5.0, Appendix V L.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. For solution (1) dilute a quantity of the nebuliser solution with sufficient of the mobile phase to produce a solution containing the equivalent of 0.05% w/v of salbutamol. For solution (2) dilute 1 volume of solution (1) to 100 volumes with the mobile phase. Solution (3) contains 0.0004% w/v of salbutamol impurity B BPCRS and 0.0005% w/v of salbutamol sulfate BPCRS in the mobile phase. For solution (1) allow the chromatography to proceed for 25 times the retention time of salbutamol (retention time of salbutamol, about 1.9 minutes).
The chromatographic procedure may be carried out using (a) a stainless steel column (15 cm × 3.9 mm) packed with spherical end-capped octylsilyl silica gel for chromatography (5 µm) with a specific surface area of 335 m2 per g, a pore size of 10 nm and a carbon loading of 11.7% (Waters Symmetry C8 is suitable), (b) as the mobile phase with a flow rate of 1 mL per minute a mixture of 22 volumes of acetonitrile and 78 volumes of a solution containing 0.287% w/v of sodium heptanesulfonate and 0.25% w/v of potassium dihydrogen orthophosphate adjusted to pH 3.65 with 2m orthophosphoric acid and (c) a detection wavelength of 220 nm.
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution factor between the two principal peaks is at least 3.0.
In the chromatogram obtained with solution (1) the area of any secondary peak is not greater than half the area of the principal peak in the chromatogram obtained with solution (2) (0.5%) and the sum of the areas of any such peaks is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1%).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions. For solution (1) dilute a quantity of the nebuliser solution, if necessary, with sufficient water to produce a solution containing the equivalent of 0.050% w/v of salbutamol. Solution (2) contains 0.00025% w/v of salbutamol ketone impurity BPCRS in water.
The chromatographic procedure may be carried out using (a) a stainless steel column (25 cm × 4.6 mm) packed with end-capped octylsilyl silica gel for chromatography (5 µm) (Hypersil BDS C8 is suitable) maintained at 30°, (b) as the mobile phase with a flow rate of 1 mL per minute the changing proportions of mobile phases A and B described below and (c) a detection wavelength of 276 nm.
Mobile phase A A mixture of 1.5 volumes of propan-2-ol and 98.5 volumes of 0.1m ammonium acetate adjusted to pH 4.5 with glacial acetic acid.
Mobile phase B propan-2-ol.
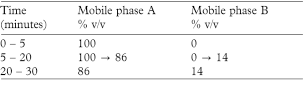
In the chromatogram obtained with solution (1) the area of any peak corresponding to salbutamol ketone is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.5%).
Dilute a volume of the nebuliser solution containing the equivalent of 4 mg of salbutamol to 50 mL with 0.1m hydrochloric acid. Measure the absorbance of the resulting solution at the maximum at 276 nm, Appendix II B, using 0.1m hydrochloric acid in the reference cell. Calculate the content of C13H21NO3 in the solution from the absorbance obtained from a solution prepared by diluting 1 volume of a 0.1% w/v solution of salbutamol sulfate BPCRS to 10 volumes with 0.1m hydrochloric acid and from the declared content of C13H21NO3 in salbutamol sulfate BPCRS.
Salbutamol Nebuliser Solution should be protected from light.
The label states the quantity of active ingredient in terms of the equivalent amount of salbutamol.