- British Pharmacopoeia Volume III
- Formulated Preparations: General Monographs
PREPARATIONS FOR INHALATION |
 |
(Ph. Eur. monograph 0671)
Preparations for Inhalation comply with the requirements of the European Pharmacopoeia. These requirements are reproduced below.
Ph Eur
Preparations for inhalation are liquid or solid preparations intended for administration as vapours or aerosols to the lung in order to obtain a local or systemic effect. They contain one or more active substances that may be dissolved or dispersed in a suitable vehicle.
Preparations for inhalation may, depending on the type of preparation, contain propellants, cosolvents, diluents, antimicrobial preservatives, solubilising and stabilising agents, etc. These excipients do not adversely affect the functions of the mucosa of the respiratory tract or its cilia.
Suspensions and emulsions are readily dispersible on shaking and they remain sufficiently stable to enable the correct dose to be delivered.
Preparations for inhalation are supplied in multidose or single-dose containers. When supplied in pressurised containers, they comply with the requirements of the monograph Pressurised pharmaceutical preparations (0523).
- — a nebuliser;
- — an inhaler (pressurised metered-dose inhaler, non-pressurised metered-dose inhaler or powder inhaler).
- — preparations to be converted into vapour;
- — liquid preparations for nebulisation;
- — pressurised metered-dose preparations for inhalation;
- — non-pressurised metered-dose preparations for inhalation;
- — inhalation powders.
During the development of a preparation for inhalation that contains an antimicrobial preservative, the effectiveness of the chosen preservative shall be demonstrated to the satisfaction of the competent authority. A suitable test method together with the criteria for judging the preservative properties of the formulation are described in general chapter 5.1.3. Efficacy of antimicrobial preservation.
In the manufacture, packaging, storage and distribution of preparations for inhalation, suitable measures are taken to ensure their microbial quality; recommendations on this aspect are provided in general chapter 5.1.4. Microbiological quality of non-sterile pharmaceutical preparations and substances for pharmaceutical use.
In assessing the uniformity of delivered dose of a multidose inhaler, it is not sufficient to test a single inhaler. Manufacturers must substitute procedures that take both inter- and intra-inhaler dose uniformity into account. A suitable procedure based on the intra-inhaler test would be to collect each of the 10 specified doses at the beginning, middle and end of the number of doses stated on the label from separate inhalers.
- — the delivered dose; alternatively, where the dose has been established as a metered dose or as a pre-dispensed dose, the label states either the metered dose or the pre-dispensed dose, as appropriate;
- — where applicable, the number of deliveries from the inhaler to provide the minimum recommended dose;
- — the number of deliveries per inhaler.
The label states, where applicable, the name of any added antimicrobial preservative.
Preparations intended to be converted into vapour are solutions, suspensions, emulsions or solid preparations. They are usually added to hot water and the vapour generated is inhaled.
Liquid preparations for nebulisation are solutions, suspensions or emulsions intended to be converted into aerosols by nebulisers.
Liquid preparations for nebulisation in concentrated form are diluted to the prescribed volume with the prescribed liquid before use. Liquid preparations for nebulisation may also be prepared from powders.
The pH of liquid preparations for nebulisation is not lower than 3 and not higher than 10.
Liquid preparations for nebulisation supplied in multidose containers may contain a suitable antimicrobial preservative at a suitable concentration except where the preparation itself has adequate antimicrobial properties.
Liquid preparations for nebulisation supplied in multidose containers that do not contain an antimicrobial preservative, and where the preparation itself does not have adequate antimicrobial properties, are sterile and are supplied in containers preventing microbial contamination of the contents during storage and use.
Liquid preparations for nebulisation supplied in single-dose containers are sterile and preservative-free, unless otherwise justified and authorised.
Nebulisers are devices that convert liquids into aerosols by high-pressure gases, ultrasonic vibration or other methods. They allow the dose to be inhaled at an appropriate active-substance delivery rate over an extended period of time involving consecutive inspirations and with a particle size that ensures deposition of the preparation in the lungs.
Nebulisers may be breath-triggered or use other means to synchronise or modify the nebuliser operation with the patient's breathing.
The active substance delivery rate and the total active substance delivered are determined using the methods described in general chapter 2.9.44. Preparations for nebulisation: characterisation. Where justified and authorised, a different apparatus and procedure may be used.
For liquid preparations for nebulisation that are solutions or suspensions, determine the particle-size distribution using an apparatus and procedure described in general chapter 2.9.44. Preparations for nebulisation: characterisation. Where justified and authorised, a different apparatus and procedure may be used.
Prepare the liquid preparation for nebulisation as directed in the instructions to the patient.
For liquid preparations for nebulisation that are suspensions, determine fine-particle mass using an apparatus and procedure described in general chapter 2.9.44. Preparations for nebulisation: characterisation. Where justified and authorised, a different apparatus and procedure may be used.
Pressurised metered-dose preparations for inhalation are solutions, suspensions or emulsions supplied in containers equipped with a metering valve and which are held under pressure with (a) suitable propellant(s), which can act also as a solvent.
The delivered dose is the dose delivered from the inhaler. For some preparations the dose has been established as a metered dose. The metered dose is determined by adding the amount deposited on the inhaler to the delivered dose. It may also be determined directly.
The size of aerosol particles to be inhaled is controlled so that a consistent portion is deposited in the lungs. The fine-particle characteristics of pressurised metered-dose preparations for inhalation are determined using the method described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles.
Pressurised metered-dose inhalers are tested for leakage.
For breath-triggered pressurised metered-dose inhalers, the test conditions described below may need to be modified to ensure that actuation occurs for the inhaler under test.
Prepare the inhaler as directed in the instructions to the patient.
Pressurised metered-dose inhalers usually operate in a valve-down position. For inhalers that operate in a valve-up position, an equivalent test is applied using methods that ensure the complete collection of the delivered dose.
The dose collection apparatus must be capable of quantitatively capturing the delivered dose.
The following apparatus (Figure 0671.-1) and procedure may be used.
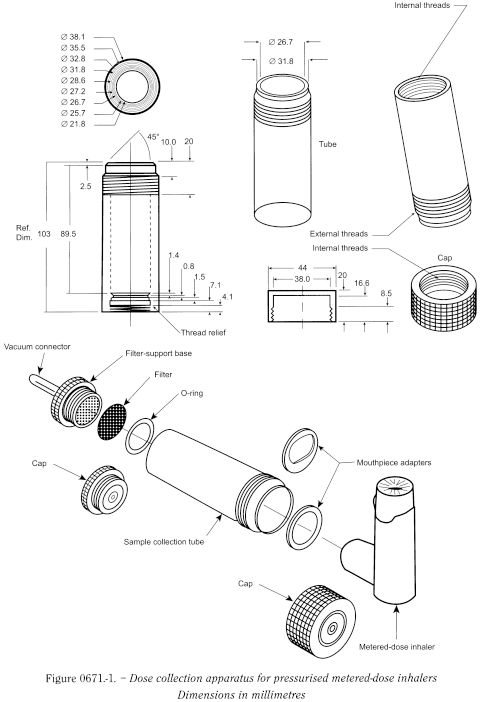
The apparatus consists of a filter-support base with an open-mesh filter-support, such as a stainless steel screen, a collection tube that is clamped or screwed to the filter-support base, and a mouthpiece adapter to ensure an airtight seal between the collection tube and the mouthpiece. Use a mouthpiece adapter that ensures that the front face of the inhaler mouthpiece is flush with the front face or the 2.5 mm indented shoulder of the sample collection tube, as appropriate. The vacuum connector is connected to a system comprising a vacuum source and a flow regulator. The source is adjusted to draw air through the complete assembly, including the filter and the inhaler to be tested, at 28.3 L/min (± 5 per cent). Air should be drawn continuously through the apparatus to avoid loss of the active substance into the atmosphere. The filter-support base is designed to accommodate 25 mm diameter filter disks. The filter disk and other materials used in the construction of the apparatus must be compatible with the active substance and solvents that are used to extract the active substance from the filter. One end of the collection tube is designed to hold the filter disk tightly against the filter-support base. When assembled, the joints between the components of the apparatus are airtight so that when a vacuum is applied to the base of the filter, all of the air drawn through the collection tube passes through the inhaler.
Unless otherwise prescribed in the instructions to the patient, shake the inhaler for 5 s and discharge 1 delivery to waste. Discharge the inverted inhaler into the apparatus, depressing the valve for a sufficient time to ensure complete discharge. Repeat the procedure until the number of deliveries that constitute the minimum recommended dose have been sampled. Quantitatively collect the contents of the apparatus and determine the amount of active substance.
Repeat the procedure for a further 2 doses.
Discharge the inhaler to waste, waiting not less than 5 s between actuations, until (n/2) + 1 deliveries remain, where n is the number of deliveries stated on the label. Collect 4 doses using the procedure described above.
Discharge the inhaler to waste, waiting not less than 5 s between actuations, until 3 doses remain. Collect these 3 doses using the procedure described above.
For preparations containing more than 1 active substance, carry out the test for uniformity of delivered dose for each active substance.
Unless otherwise justified and authorised, the preparation complies with the test if 9 out of 10 results lie between 75 per cent and 125 per cent of the average value and all lie between 65 per cent and 135 per cent. If 2 or 3 values lie outside the limits of 75 per cent to 125 per cent, repeat the test for 2 more inhalers. Not more than 3 of the 30 values lie outside the limits of 75 per cent to 125 per cent and no value lies outside the limits of 65 per cent to 135 per cent.
Using an apparatus and procedure described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles (apparatus C, D or E), calculate the fine particle dose.
Take 1 inhaler and discharge the contents to waste, actuating the valve at intervals of not less than 5 s. The total number of deliveries so discharged from the inhaler is not less than the number stated on the label (this test may be combined with the test for uniformity of delivered dose).
Non-pressurised metered-dose preparations for inhalation are solutions, suspensions or emulsions for use with inhalers that convert liquids into aerosols using single or multiple liquid jets, ultrasonic vibration or other methods. The volume of liquid to be converted into an aerosol is pre-metered or metered by the inhaler so that the dose delivered from the inhaler can be inhaled with 1 or more inspirations.
Non-pressurised metered-dose preparations for inhalation supplied in multidose containers may contain a suitable antimicrobial preservative at a suitable concentration except where the preparation itself has adequate antimicrobial properties.
Non-pressurised metered-dose preparations for inhalation supplied in multidose containers that do not contain an antimicrobial preservative, and where the preparation itself does not have adequate antimicrobial properties, are sterile and are supplied in containers preventing microbial contamination of the contents during storage and use.
Non-pressurised metered-dose preparations for inhalation supplied in single-dose containers are sterile and preservative-free, unless otherwise justified and authorised.
The size of aerosol particles to be inhaled is controlled so that a consistent portion is deposited in the lung. The fine-particle characteristics of non-pressurised metered-dose preparations for inhalation are determined using the method described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles. Alternatively, laser diffraction analysis may be used, when properly validated against method 2.9.18 (apparatus C, D or E).
For breath-triggered non-pressurised metered-dose inhalers, the test conditions described below may need to be modified to ensure that actuation occurs for the inhaler under test.
Prepare the inhaler as directed in the instructions to the patient.
The dose collection apparatus must be capable of quantitatively capturing the delivered dose. The apparatus described in the test for uniformity of delivered dose for pressurised metered-dose preparations may be used.
Discharge the inhaler into the apparatus. Repeat the procedure until the number of deliveries that constitute the minimum recommended dose have been sampled. Quantitatively collect the contents of the apparatus and determine the amount of active substance.
Repeat the procedure for a further 2 doses.
Discharge the inhaler to waste until (n/2) + 1 deliveries remain, where n is the number of deliveries stated on the label. Collect 4 doses using the procedure described above.
Discharge the inhaler to waste until 3 doses remain. Collect these 3 doses using the procedure described above.
For preparations containing more than 1 active substance, carry out the test for uniformity of delivered dose for each active substance.
Unless otherwise justified and authorised, the preparation complies with the test if 9 out of 10 results lie between 75 per cent and 125 per cent of the average value and all lie between 65 per cent and 135 per cent. If 2 or 3 values lie outside the limits of 75 per cent to 125 per cent, repeat the test for 2 more inhalers. Not more than 3 of the 30 values lie outside the limits of 75 per cent to 125 per cent and no value lies outside the limits of 65 per cent to 135 per cent.
Where justified and authorised, another apparatus and procedure may be used.
Using an apparatus and procedure described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles (apparatus C, D or E), calculate the fine particle dose. Use the same procedure as for pressurised inhalers with appropriate adaptation of the methodology to non-pressurised inhalers. Depending on the characteristics of the non-pressurised metered-dose preparations for inhalation, relative humidity and/or temperature may need to be controlled during the test.
Take 1 inhaler and discharge the contents to waste. The total number of deliveries so discharged from the inhaler is not less than the number stated on the label (this test may be combined with the test for uniformity of delivered dose).
Inhalation powders are supplied in single-dose or multidose containers. To facilitate their use, active substances may be combined with a suitable carrier. They are administered by powder inhalers. For pre-metered inhalers, the inhaler is loaded with powders pre-dispensed in capsules or other suitable dosage forms. For inhalers using a powder reservoir, the dose is created by a metering mechanism within the inhaler.
The delivered dose is the dose delivered from the inhaler. For some preparations, the labelled dose has been established as a metered dose or as a pre-dispensed dose. The metered dose is determined by adding the amount deposited on the inhaler to the delivered dose. It may also be determined directly.
The size of aerosol particles to be inhaled is controlled so that a consistent portion is deposited in the lung. The fine-particle characteristics of powders for inhalation are determined using the method described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles.
Prepare the inhaler as directed in the instructions to the patient.
The dose collection apparatus must be capable of quantitatively capturing the delivered dose. A dose collection apparatus similar to that described for the evaluation of pressurised metered-dose inhalers may be used provided that the dimensions of the tube and the filter can accommodate the measured flow rate. A suitable tube is defined in Table 0671.-1. Connect the tube to a flow system according to the scheme specified in Figure 0671.-2 and Table 0671.-1.
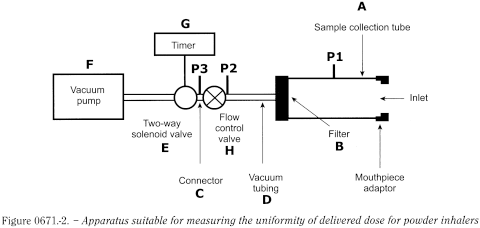
Unless otherwise stated, determine the test flow rate and duration using the dose collection tube, the associated flow system, a suitable differential pressure meter and a suitable volumetric flowmeter, calibrated for the flow leaving the meter, according to the following procedure.
Prepare the inhaler for use and connect it to the inlet of the apparatus using a mouthpiece adapter to ensure an airtight seal. Use a mouthpiece adapter that ensures that the front face of the inhaler mouthpiece is flush with the front face of the sample collection tube. Connect one port of a differential pressure meter to the pressure reading point P1 in Figure 0671.-2, and let the other be open to the atmosphere. Switch on the pump, open the 2-way solenoid valve and adjust the flow control valve until the pressure drop across the inhaler is 4.0 kPa (40.8 cm H2O) as indicated by the differential pressure meter. Remove the inhaler from the mouthpiece adapter and, without touching the flow control valve, connect a flowmeter to the inlet of the sampling apparatus. Use a flowmeter calibrated for the volumetric flow leaving the meter, or calculate the volumetric flow leaving the meter (Qout) using the ideal gas law. For a meter calibrated for the entering volumetric flow (Qin), use the following expression:
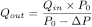
P0 |
= |
atmospheric pressure, |
ΔP |
= |
pressure drop over the meter. |
If the flow rate is above 100 L/min adjust the flow control valve to obtain a flow rate of 100 L/min (± 5 per cent). Note the volumetric airflow rate exiting the meter and define this as the test flow rate, Qout, in litres per minute. Define the test flow duration, T, in seconds so that a volume of 4 L of air is drawn from the mouthpiece of the inhaler at the test flow rate, Qout.
Ensure that critical flow occurs in the flow control valve by the following procedure: with the inhaler in place and the test flow rate Qout, measure the absolute pressure on both sides of the control valve (pressure reading points P2 and P3 in Figure 0671.-2); a ratio P3/P2 of less than or equal to 0.5 indicates critical flow; switch to a more powerful pump and re-measure the test flow rate if critical flow is not indicated.
Pre-dispensed systems Connect the inhaler to the apparatus using an adapter that ensures a good seal. Draw air through the inhaler using the predetermined conditions. Repeat the procedure until the number of deliveries that constitute the minimum recommended dose have been sampled. Quantitatively collect the contents of the apparatus and determine the amount of active substance.
Repeat the procedure for a further 9 doses.
Reservoir systems Connect the inhaler to the apparatus using an adapter that ensures a good seal. Draw air through the inhaler under the predetermined conditions. Repeat the procedure until the number of deliveries that constitute the minimum recommended dose have been sampled. Quantitatively collect the contents of the apparatus and determine the amount of active substance.
Repeat the procedure for a further 2 doses.
Discharge the inhaler to waste until (n/2) + 1 deliveries remain, where n is the number of deliveries stated on the label. If necessary, store the inhaler to discharge electrostatic charges. Collect 4 doses using the procedure described above.
Discharge the inhaler to waste until 3 doses remain. If necessary, store the inhaler to discharge electrostatic charges. Collect 3 doses using the procedure described above.
For preparations containing more than 1 active substance, carry out the test for uniformity of delivered dose for each active substance.
Results The preparation complies with the test if 9 out of 10 results lie between 75 per cent and 125 per cent of the average value and all lie between 65 per cent and 135 per cent. If 2 or 3 values lie outside the limits of 75 per cent to 125 per cent, repeat the test for 2 more inhalers. Not more than 3 of the 30 values lie outside the limits of 75 per cent to 125 per cent and no value lies outside the limits of 65 per cent to 135 per cent.
In justified and authorised cases, these ranges may be extended but no value should be greater than 150 per cent or less than 50 per cent of the average value.
Using an apparatus and procedure described in general chapter 2.9.18. Preparations for inhalation: aerodynamic assessment of fine particles (apparatus C, D or E), calculate the fine particle dose.
Discharge doses from the inhaler until empty, at the predetermined flow rate. Record the deliveries discharged. The total number of deliveries so discharged from the inhaler is not less than the number stated on the label (this test may be combined with the test for uniformity of delivered dose).
Ph Eur