- British Pharmacopoeia Volume III
- Formulated Preparations: Specific Monographs
Salbutamol Pressurised Inhalation |
Beta2-adrenoceptor agonist; bronchodilator.
Salbutamol Pressurised Inhalation is a suspension of either Salbutamol or Salbutamol Sulfate in a suitable liquid in a suitable pressurised container.
The pressurised inhalation complies with the requirements stated under Preparations for Inhalation and with the following requirements.
80.0 to 120.0% of the amount stated to be delivered by actuation of the valve.
A. The infrared absorption spectrum, Appendix II A, in the range 1650 to 400 cm-1 is concordant with the reference spectrum of either salbutamol (RS 314) or salbutamol sulfate (RS 315), as appropriate. Examine the substance as a dispersion in potassium bromide prepared in the following manner. Discharge the inhaler a sufficient number of times into a mortar to obtain 2 mg of Salbutamol, grind the residue thoroughly with 0.1 g of potassium bromide, add a further 0.2 g of potassium bromide and mix thoroughly.
B. In the test for Related substances, the principal peak in the chromatogram obtained with solution (2) corresponds to the peak due to salbutamol in the chromatogram obtained with solution (3).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in the mobile phase.
(1) Discharge the Salbutamol inhaler or the Salbutamol Sulfate inhaler as appropriate, a sufficient number of times into a small, dry beaker to obtain 5 mg of Salbutamol and dissolve the residue in 10 mL of the mobile phase; mix with the aid of ultrasound until the solution is clear.
(2) Dilute 1 volume of solution (1) to 100 volumes.
(3) 0.0004% w/v of salbutamol impurity B BPCRS and 0.0005% w/v of salbutamol sulfate BPCRS.
(a) Use a stainless steel column (15 cm × 3.9 mm) packed with spherical end-capped octylsilyl silica gel for chromatography (5 µm) with a specific surface area of 335 m2 per g, a pore size of 10 nm and a carbon loading of 11.7% (Waters Symmetry C8 is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 1 mL per minute.
(d) Use an ambient column temperature.
(e) Use a detection wavelength of 220 nm.
(f) Inject 20 µL of each solution.
(g) For solution (1) allow the chromatography to proceed for 25 times the retention time of salbutamol (retention time of salbutamol, about 1.9 minutes).
22 volumes of acetonitrile and 78 volumes of a solution containing 0.287% w/v of sodium heptanesulfonate and 0.25% w/v of potassium dihydrogen orthophosphate adjusted to pH 3.65 with 2m orthophosphoric acid.
The test is not valid unless, in the chromatogram obtained with solution (3), the resolution factor between the two principal peaks is at least 3.0.
In the chromatogram obtained with solution (1):
the area of any secondary peak is not greater than half the area of the principal peak in the chromatogram obtained with solution (2) (0.5%);
the sum of the areas of any such peaks is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (1%).
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in water.
(1) Dilute a quantity of the contents of the inhaler with sufficient water to produce a solution containing the equivalent of 0.050% w/v of Salbutamol.
(2) 0.00025% w/v of salbutamol ketone impurity BPCRS.
(a) Use a stainless steel column (25 cm × 4.6 mm) packed with end-capped octylsilyl silica gel for chromatography (5 µm) (Hypersil BDS C8 is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 1 mL per minute.
(d) Use a column temperature of 30°.
(e) Use a detection wavelength of 276 nm.
(f) Inject 20 µL of each solution.
Mobile phase A A mixture of 1.5 volumes of propan-2-ol and 98.5 volumes of 0.1m ammonium acetate adjusted to pH 4.5 with glacial acetic acid.
Mobile phase B propan-2-ol.
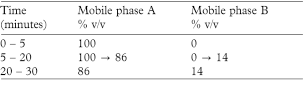
In the chromatogram obtained with solution (1):
the area of any peak corresponding to salbutamol ketone is not greater than the area of the principal peak in the chromatogram obtained with solution (2) (0.5%).
Carry out the test for aerodynamic assessment of fine particles, Appendix XII C7, using Apparatus A but determining the content of active ingredient as described below.
For pressurised inhalations containing Salbutamol use 7 mL of methanol in the upper impingement chamber and 30 mL of methanol in the lower impingement chamber. Use methanol to wash the coupling tube, E, and transfer the combined solution and washings in the lower impingement chamber to a 100 mL graduated flask containing 22.5 mL of a 0.1% w/v solution of ammonium acetate, rinsing the chamber with methanol, and dilute the combined solution and washings to 100 mL with methanol.
For pressurised inhalations containing Salbutamol Sulfate use 7 mL of water in the upper impingement chamber and 30 mL of water in the lower impingement chamber. Use a mixture of equal volumes of methanol and water to wash the coupling tube, E, and transfer the combined solution and washings in the lower impingement chamber to a 100 mL graduated flask containing 22.5 mL of a 0.1% w/v solution of ammonium acetate, rinsing the chamber with a mixture of equal volumes of methanol and water, and dilute the combined solution and washings to 100 mL with water.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in the mobile phase.
(1) 0.0006% w/v of 2-tert-butylamino-1-(4-hydroxy-3-methylphenyl)ethanol sulfate BPCRS and 0.0005% w/v of salbutamol BPCRS.
(2) 0.0005% w/v solution of salbutamol BPCRS.
(3) Use the diluted solution from the lower impingement chamber.
(a) Use a stainless steel column 10 cm × 5 mm) packed with octadecylsilyl silica gel for chromatography (5 µm) (Spherisorb ODS 1 is suitable).
(b) Use isocratic elution and the mobile phase described below.
(c) Use a flow rate of 2 mL per minute.
(d) Use an ambient column temperature.
(e) Use a detection wavelength of 276 nm.
(f) Inject 20 µL of each solution.
225 volumes of a 0.1% w/v solution of ammonium acetate and 800 volumes of methanol.
The test is not valid unless the resolution factor between the two principal peaks in the chromatogram obtained with solution (1) is at least 1.5.
Calculate the amount of salbutamol, C13H21NO3, delivered to the lower impingement chamber per actuation of the valve using the declared content of C13H21NO3 in salbutamol BPCRS. Not less than 35% of the average amount of salbutamol delivered per actuation of the valve, calculated as the average of the three results determined in the Assay, is deposited in the lower impingement chamber.
Determine the content of active ingredient delivered by the first 10 successive combined actuations of the valve after priming.
For pressurised inhalations containing Salbutamol carry out the procedure for Content of active ingredient delivered by actuation of the valve described under Pressurised Inhalations, beginning at the words 'Remove the pressurised container from the actuator…' and ending at the words '… to the volume specified in the monograph', using 35 mL of methanol in the vessel. Add 22.5 mL of a 0.1% w/v solution of ammonium acetate to the combined solution and washings obtained from the set of 10 combined actuations and dilute to 100 mL with methanol (solution A).
For pressurised inhalations containing Salbutamol Sulfate carry out the procedure for Content of active ingredient delivered by actuation of the valve described under Pressurised Inhalations, beginning at the words 'Remove the pressurised container from the actuator…' and ending at the words '… to the volume specified in the monograph', using 35 mL of a mixture of equal volumes of methanol and water in the vessel. Add 22.5 mL of a 0.1% w/v solution of ammonium acetate to the combined solution and washings obtained from the set of 10 combined actuations and dilute to 100 mL with water (solution A).
Determine the amount of active ingredient in the 10 combined actuations using the following method of analysis.
Carry out the method for liquid chromatography, Appendix III D, using the following solutions in the mobile phase.
(1) 0.0012% w/v of 2-tert-butylamino-1-(4-hydroxy-3-methylphenyl)ethanol sulfate BPCRS and 0.001% w/v of salbutamol BPCRS.
(2) 0.001% w/v of salbutamol BPCRS.
(3) Use solution A.
The chromatographic conditions described under Deposition of the emitted dose may be used.
The test is not valid unless the resolution factor between the two principal peaks in the chromatogram obtained with solution (1) is at least 1.5.
Calculate the average content of C13H21NO3 delivered by a single actuation of the valve using the declared content of C13H21NO3 in salbutamol BPCRS.
Determine the content of active ingredient a second and a third time by repeating the procedure on the middle 10 and on the last 10 successive combined actuations of the valve, as estimated from the number of deliveries available from the container as stated on the label. For each of the three determinations the average content of C13H21NO3 delivered by a single actuation of the valve is within the limits stated under Content of salbutamol.
The label on the container states (1) whether the preparation contains Salbutamol or Salbutamol Sulfate; (2) where applicable, that the preparation does not contain chlorofluorocarbon propellants (CFCs).
When the active ingredient is Salbutamol Sulfate, the quantity is stated in terms of the equivalent amount of salbutamol.