| 参考文献No. | 694592 |
| 标题: | Novel antimicrobial pyridazine phenyloxazolidinones |
| 作者: | Luehr, G.W.; Wang, S.; Hackbarth, C.J.; Gomez, M.; Trias, J.; Patel, D.V.; Gordeev, M.F. |
| 来源: | 42nd Intersci Conf Antimicrob Agents Chemother (Sept 27 2002, San Diego) 2002,Abst F-1320 |
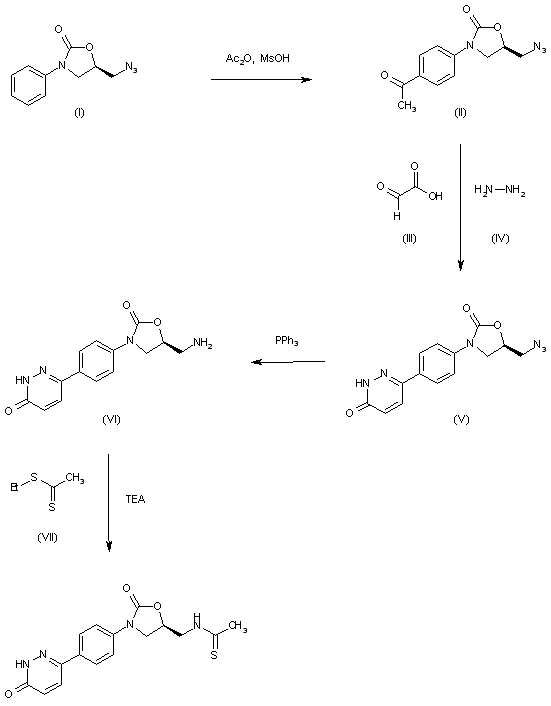 |
| 合成路线图解说明: The reaction of 5(R)-(azidomethyl)-3-phenyloxazolidin-2-one (I) with Ac2O and MsOH gives the 4-acetylphenyl derivative (II), which is cyclized with glyoxylic acid (III) and hydrazine (IV) in hot acetic acid to yield the pyridazinone derivative (V). The reduction of the azido group of (V) by means of PPh3 in THF/water affords the aminomethyl compound (VI), which is finally condensed with ethyl dithioacetate (VII) by means of TEA in DMF to provide the target thioacetamide. |
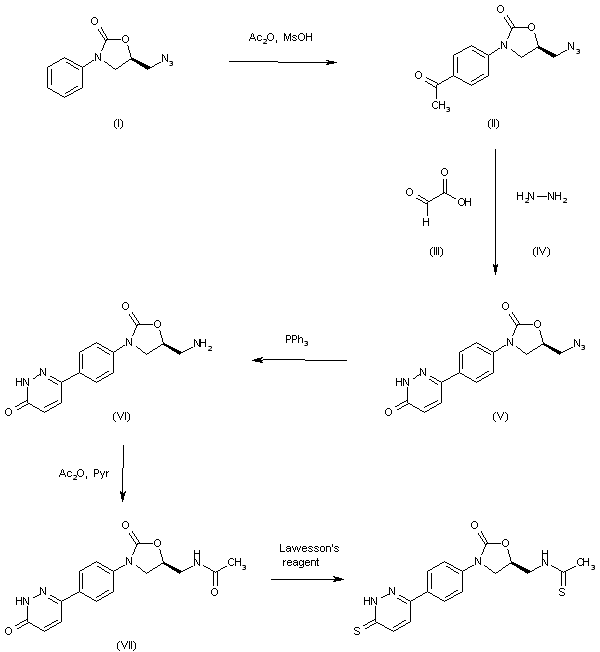 |
| 合成路线图解说明: The reaction of 5(R)-(azidomethyl)-3-phenyloxazolidin-2-one (I) with Ac2O and MsOH gives the 4-acetylphenyl derivative (II), which is cyclized with glyoxylic acid (III) and hydrazine (IV) in hot acetic acid to yield the pyridazinone derivative (V). The reduction of the azido group of (V) by means of PPh3 in THF/water affords the aminomethyl compound (VI), which is condensed with acetic anhydride and pyridine to provide the acetamide (VII). Finally, this compound is treated with Lawesson's reagent in hot dioxane to furnish the target pyridazinethione. |
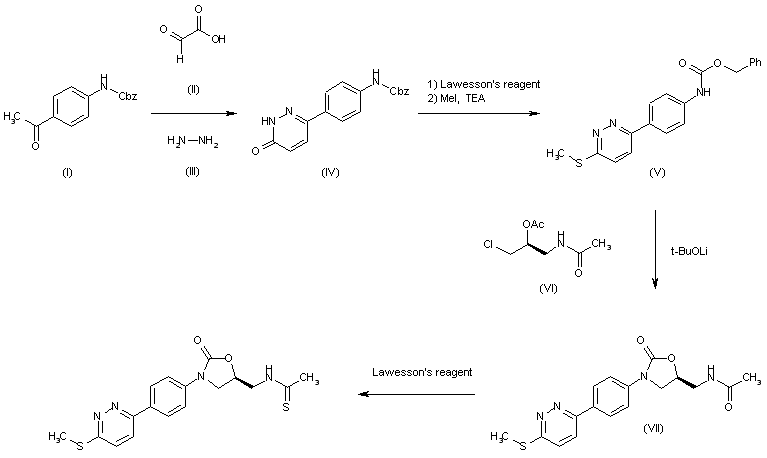 |
| 合成路线图解说明: The cyclization of 4-(benzyloxycarbonylamino)acetophenone (I) with glyoxylic acid (II) and hydrazine (III) in hot acetic acid gives the pyridazinone (IV), which is treated first with Lawesson's reagent in hot dioxane and then with MeI and TEA in DMF to yield the methylsulfanyl pyridazine (V). The cyclization of (V) with acetamide (VI) by means of t-BuOLi in methanol/DMF affords the oxazolidinone (VII), which is finally treated with Lawesson's reagent in hot dioxane to provide the target thioacetamide. |
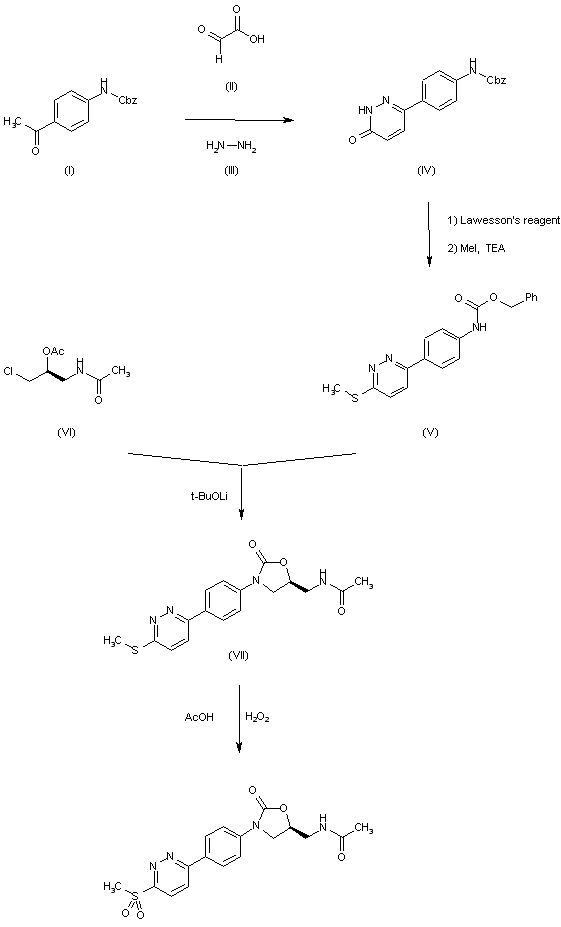 |
| 合成路线图解说明: The cyclization of 4-(benzyloxycarbonylamino)acetophenone (I) with glyoxylic acid (II) and hydrazine (III) in hot acetic acid gives the pyridazinone (IV), which is treated first with Lawesson's reagent in hot dioxane and then with MeI and TEA in DMF to yield the methylsulfanyl pyridazine (V). The cyclization of (V) with acetamide (VI) by means of t-BuOLi in methanol/DMF affords the oxazolidinone (VII), which is finally treated with Lawesson's reagent in hot dioxane to provide the target thioacetamide. |