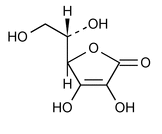
Ascorbic Acid
(as kore' bik as' id).
DEFINITION
Ascorbic Acid contains NLT 99.0% and NMT 100.5% of C6H8O6.
IDENTIFICATION
• B. A 20-mg/mL solution reduces alkaline cupric tartrate TS slowly at room temperature but more readily upon heating.
ASSAY
• Procedure
Sample: 400 mg of Ascorbic Acid
Titrimetric system
(See Titrimetry  541
541 .)
.)
Mode: Direct titration
Titrant: 0.1 N iodine VS
Endpoint detection: Visual
Blank: 100 mL of water and 25 mL of 2 N sulfuric acid. Add 3 mL of starch TS.
Analysis: Dissolve the Sample in a mixture of 100 mL of water and 25 mL of 2 N sulfuric acid. Add 3 mL of starch TS, and titrate immediately with Titrant until a persistent violet-blue color is obtained.
Calculate the percentage of ascorbic acid (C6H8O6) in the portion of Ascorbic Acid taken:
Result = [(V  B) × N × F × 100]/W
B) × N × F × 100]/W
| V | = | = sample titrant volume (mL) |
| B | = | = blank titrant volume (mL) |
| N | = | = titrant normality (mEq/mL) |
| F | = | = equivalency factor, 88.06 mg/mEq |
| W | = | = weight of Sample (mg) |
Acceptance criteria: 99.0%–100.5%
IMPURITIES
• Residue on Ignition  281
281 : NMT 0.1%
: NMT 0.1%
SPECIFIC TESTS
• Optical Rotation, Specific Rotation  781S
781S
Sample solution: 100 mg/mL in carbon dioxide-free water. Perform the test immediately after preparation of the Sample solution.
Acceptance criteria: +20.5 to +21.5
to +21.5
ADDITIONAL REQUIREMENTS
• Packaging and Storage: Preserve in tight, light-resistant containers.
Auxiliary Information— Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Huy T. Dinh, M.S. Scientific Liaison 1-301-816-8594 | (DS2010) Monographs - Dietary Supplements |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2242
Chromatographic Column—
Chromatographic columns text is not derived from, and not part of, USP 35 or NF 30.