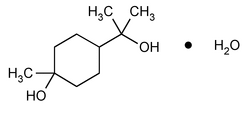Terpin Hydrate
(ter' pin hye' drate).
Cyclohexanemethanol, 4-hydroxy-
p-Menthane-1,8-diol monohydrate
Anhydrous 172.27
» Terpin Hydrate contains not less than 98.0 percent and not more than 100.5 percent of C10H20O2, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight containers.
USP Reference standards  11
11 —
—
USP Terpin Hydrate RS
Identification—
A:
Infrared Absorption  197K
197K : dried over a suitable desiccant for 4 hours.
: dried over a suitable desiccant for 4 hours.
B:
Add a few drops of sulfuric acid to a hot solution: the liquid becomes turbid and develops a strongly aromatic odor.
Water, Method I  921
921 :
between 9.0% and 10.0%.
:
between 9.0% and 10.0%.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Residual turpentine—
It has no odor of turpentine oil.
Assay—
Internal standard solution—
Dissolve a quantity of biphenyl in chloroform to obtain a solution containing about 20 mg per mL.
Standard preparation—
Transfer about 170 mg of USP Terpin Hydrate RS, accurately weighed, to a 100-mL volumetric flask, dissolve in 5 mL of alcohol, add 5.00 mL of Internal standard solution, dilute with chloroform to volume, and mix.
Assay preparation—
Transfer about 170 mg of Terpin Hydrate, accurately weighed, to a 100-mL volumetric flask, and proceed as directed under Standard preparation, beginning with “dissolve in 5 mL of alcohol.”
Chromatographic system—
Under typical conditions, the gas chromatograph is equipped with a flame-ionization detector, and it contains a 3.5-mm × 1.2-m glass column packed with 6% G1 on support S1A; the injection port and the detector temperatures are maintained at 260 , and the column temperature at 120
, and the column temperature at 120 ; and nitrogen is used as the carrier gas, at a flow rate necessary to yield approximate retention times of 7 minutes for terpin and 11 minutes for biphenyl.
; and nitrogen is used as the carrier gas, at a flow rate necessary to yield approximate retention times of 7 minutes for terpin and 11 minutes for biphenyl.
System suitability test—
Chromatograph the Standard preparation, and record the chromatograms as directed for Procedure: the resolution, R, between the terpin and biphenyl peaks is not less than 2.0, and the relative standard deviation for replicate injections is not more than 2.0%.
Procedure—
Inject about 1 µL of the Standard preparation into a suitable gas chromatograph, and record the chromatogram. Similarly, inject about 1 µL of the Assay preparation, and record the chromatogram. Calculate the quantity, in mg, of C10H20O2 in the portion of Terpin Hydrate taken by the formula:
WS(RU / RS)
in which WS is the weight, in mg, of USP Terpin Hydrate RS, calculated on the anhydrous basis; and RU and RS are the area-ratios of terpin to the internal standard obtained from the chromatograms for the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4797