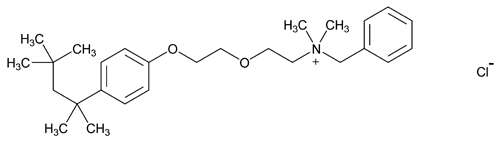
Benzethonium Chloride
C27H42ClNO2 448.08
Benzenemethanaminium, N,N-dimethyl-N-[2-[2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-, chloride;
Benzyldimethyl[2-[2-[p-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]ammonium chloride

 [121-54-0].
[121-54-0].
(ben'' ze thoe' nee um klor' ide).
C27H42ClNO2 448.08
Benzenemethanaminium, N,N-dimethyl-N-[2-[2-[4-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]-, chloride;
Benzyldimethyl[2-[2-[p-(1,1,3,3-tetramethylbutyl)phenoxy]ethoxy]ethyl]ammonium chloride
DEFINITION
Benzethonium Chloride contains NLT 97.0% and NMT 103.0% of C27H42ClNO2, calculated on the dried basis.
IDENTIFICATION
• A. Procedure
Sample solution:
10 mg/mL
Analysis:
To 1 mL of the Sample solution add 2 mL of alcohol, 0.5 mL of 2 N nitric acid, and 1 mL of silver nitrate TS.
Acceptance criteria:
A white precipitate, which is insoluble in 2 N nitric acid but soluble in 6 N ammonium hydroxide, is formed.
ASSAY
• Procedure
Sample:
0.3 g of Benzethonium Chloride
Analysis:
Dissolve the Sample in 75 mL of water contained in a glass-stoppered, 250-mL flask. Add 0.4 mL of bromophenol blue solution (1 in 2000), 10 mL of chloroform, and 1 mL of 1 N sodium hydroxide. Titrate with 0.02 M sodium tetraphenylboron VS until the blue color disappears from the chloroform layer. Add the last portions of the sodium tetraphenylboron solution dropwise, agitating vigorously after each addition. Each mL of 0.02 M sodium tetraphenylboron is equivalent to 8.962 mg of C27H42ClNO2.
Acceptance criteria:
97.0%–103.0% on the dried basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%
:
NMT 0.1%
Organic Impurities
• Procedure: Limit of Ammonium Compounds:
To 5 mL of a solution (1 in 50) add 3 mL of 1 N sodium hydroxide, and heat to boiling: the odor of ammonia is not perceptible.
SPECIFIC TESTS
• Melting Range or Temperature  741
741 :
158
:
158 –163
–163 , the specimen having been dried previously
, the specimen having been dried previously
• Loss on Drying  731
731 :
Dry a sample at 105
:
Dry a sample at 105 for 4 h: it loses NMT 5.0% of its weight.
for 4 h: it loses NMT 5.0% of its weight.
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientific Liaison 1-301-816-8394 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2317
Pharmacopeial Forum: Volume No. 35(4) Page 833