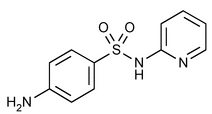Sulfapyridine
(sul'' fa pir' i deen).
Benzenesulfonamide, 4-amino-N-2-pyridinyl-.
N1-2-Pyridylsulfanilamide
» Sulfapyridine contains not less than 99.0 percent and not more than 100.5 percent of C11H11N3O2S, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Clarity and color of solution—
A solution of 1.0 g in a mixture of 20 mL of water and 5 mL of 1 N sodium hydroxide is clear and not more deeply colored than pale yellow.
Identification—
B:
Add 5 mL of 3 N hydrochloric acid to about 0.1 g of Sulfapyridine, and boil gently for about 5 minutes. Cool in an ice bath, add 4 mL of sodium nitrite solution (1 in 100), dilute with water to 10 mL, and place the mixture in the ice bath for 10 minutes. To 5 mL of the cooled mixture add a solution of 50 mg of 2-naphthol in 2 mL of sodium hydroxide solution (1 in 10): an orange-red precipitate is formed, and it darkens on standing.
Melting range  741
741 :
between 190
:
between 190 and 193
and 193 .
.
Acidity—
Digest 2.0 g with 100 mL of water at about 70 for 5 minutes, cool at once to about 20
for 5 minutes, cool at once to about 20 , and filter. To 25.0 mL of the filtrate add 2 drops of phenolphthalein TS, and titrate with 0.10 N sodium hydroxide: not more than 0.20 mL is required for neutralization.
, and filter. To 25.0 mL of the filtrate add 2 drops of phenolphthalein TS, and titrate with 0.10 N sodium hydroxide: not more than 0.20 mL is required for neutralization.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 2 hours: it loses not more than 0.5% of its weight.
for 2 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Selenium  291
291 :
0.003%, a 200-mg test specimen being used.
:
0.003%, a 200-mg test specimen being used.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Assay—
Proceed with Sulfapyridine as directed under Nitrite Titration  451
451 . Each mL of 0.1 N sodium nitrite is equivalent to 24.93 mg of C11H11N3O2S.
. Each mL of 0.1 N sodium nitrite is equivalent to 24.93 mg of C11H11N3O2S.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Leonel M. Santos, Ph.D.
Senior Scientific Liaison 1-301-816-8168 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4720