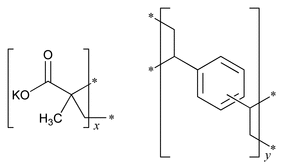Polacrilin Potassium
(pol'' a kril' in poe tas' ee um).
2-Propenoic acid, 2-methyl-, potassium salt, polymer with diethenylbenzene.
Potassium methacrylate-divinylbenzene copolymer
» Polacrilin Potassium is the potassium salt of a unifunctional low-cross-linked carboxylic cation-exchange resin prepared from methacrylic acid and divinylbenzene. When previously dried at 105 for 6 hours, it contains not less than 20.6 percent and not more than 25.1 percent of potassium.
for 6 hours, it contains not less than 20.6 percent and not more than 25.1 percent of potassium.
Packaging and storage—
Preserve in well-closed containers.
Identification—
B:
Shake about 1 g with 10 mL of water: the aqueous phase does not respond to the tests for Potassium  191
191 . Shake about 1 g with 10 mL of 0.1 N hydrochloric acid: the aqueous phase responds to the tests for Potassium
. Shake about 1 g with 10 mL of 0.1 N hydrochloric acid: the aqueous phase responds to the tests for Potassium  191
191 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 6 hours: it loses not more than 10.0% of its weight.
for 6 hours: it loses not more than 10.0% of its weight.
Powder fineness  811
811 —
Transfer about 4 g, accurately weighed, to a No. 100 standard sieve placed on top of a No. 200 standard sieve and pan. Using a soft 2-cm brush, brush the sample lightly across the No. 100 sieve until no more particles pass through. By brushing and tapping, dust off the particles on the underside of the No. 100 sieve into the No. 200 sieve. Obtain the weight of the material retained on the No. 100 sieve. Similarly, determine the weight of material retained by the No. 200 sieve; not more than 1.0% is retained on the No. 100 sieve, and not more than 30.0% is retained on the No. 200 sieve.
—
Transfer about 4 g, accurately weighed, to a No. 100 standard sieve placed on top of a No. 200 standard sieve and pan. Using a soft 2-cm brush, brush the sample lightly across the No. 100 sieve until no more particles pass through. By brushing and tapping, dust off the particles on the underside of the No. 100 sieve into the No. 200 sieve. Obtain the weight of the material retained on the No. 100 sieve. Similarly, determine the weight of material retained by the No. 200 sieve; not more than 1.0% is retained on the No. 100 sieve, and not more than 30.0% is retained on the No. 200 sieve.
Iron  241
241 —
Transfer 0.10 g to a suitable crucible, and ignite at a low heat until thoroughly ashed. Add to the carbonized mass 2 mL of nitric acid and 5 drops of sulfuric acid, and heat cautiously until white fumes are no longer evolved. Ignite, preferably in a muffle furnace, at 500
—
Transfer 0.10 g to a suitable crucible, and ignite at a low heat until thoroughly ashed. Add to the carbonized mass 2 mL of nitric acid and 5 drops of sulfuric acid, and heat cautiously until white fumes are no longer evolved. Ignite, preferably in a muffle furnace, at 500 to 600
to 600 , until the carbon is completely burned off. Cool, add 4 mL of 6 N hydrochloric acid, cover, digest on a steam bath for 15 minutes, uncover, and slowly evaporate on a steam bath to dryness. Moisten the residue with 1 drop of hydrochloric acid, add 10 mL of hot water, and digest for 2 minutes. Dilute with water to about 25 mL. Filter, if necessary, rinse the crucible and the filter with 10 mL of water, combining the filtrate and rinsing in a 50-mL color-comparison tube, add 2 mL of hydrochloric acid, dilute with water to 45 mL, and mix. The limit is 0.01%.
, until the carbon is completely burned off. Cool, add 4 mL of 6 N hydrochloric acid, cover, digest on a steam bath for 15 minutes, uncover, and slowly evaporate on a steam bath to dryness. Moisten the residue with 1 drop of hydrochloric acid, add 10 mL of hot water, and digest for 2 minutes. Dilute with water to about 25 mL. Filter, if necessary, rinse the crucible and the filter with 10 mL of water, combining the filtrate and rinsing in a 50-mL color-comparison tube, add 2 mL of hydrochloric acid, dilute with water to 45 mL, and mix. The limit is 0.01%.
Sodium—
Test solution—
Transfer about 2 g, accurately weighed, to a 400-mL borosilicate beaker, add 20 mL of sulfuric acid, cover with a borosilicate watch glass, and heat until charring is complete. While continuing to heat the beaker, add 20 mL of nitric acid dropwise. Continue to heat and add nitric acid until all of the organic material has been destroyed as indicated by the contents of the beaker turning from brown to a very pale straw-colored or colorless solution. Continue to evaporate the solution, and if it turns brown during the evaporation, add nitric acid dropwise until the brown color disappears. Evaporate just to dryness, cool, and dissolve the residue in 40 mL of water and 10 mL of 6 N hydrochloric acid. Heat to boiling, cool, transfer to a 100-mL volumetric flask, dilute with water to volume, and mix.
Procedure—
To three separate 100-mL volumetric flasks add, respectively, 1.00 mL, 2.00 mL, and 3.00 mL of a solution containing 254.2 mg of sodium chloride in 1000 mL of water. Add water to volume, and mix to obtain sodium chloride solutions having concentrations equivalent to 1 µg of Na per mL, 2 µg of Na per mL, and 3 µg of Na per mL, respectively. Adjust the settings of a suitable flame photometer so that the emission of the solution containing 3.00 mL of the sodium chloride solution reads close to 100% at 589 nm. Determine the readings of the three solutions at 589 nm. Readjust the wavelength setting to 580 nm, and determine the background emission reading for one of these standards. Pipet 5 mL of the Test solution into a 100-mL volumetric flask, add water to volume, and mix. Observe the emission reading of this solution at 589 nm, using the same instrument settings, then readjust the wavelength setting to 580 nm, and observe the background emission reading. Subtract the corresponding background readings from the standard and test specimen readings. Prepare a standard curve by plotting the corrected standard readings versus the square root of the sodium concentration. From this standard curve, determine the sodium content of the test specimen. It is not greater than 0.20%.
Heavy metals, Method III  231
231 :
0.002%.
:
0.002%.
Assay for potassium—
Sodium stock solution—
Transfer 7.306 g of sodium chloride, previously dried at 125 for 30 minutes and accurately weighed, to a 500-mL volumetric flask, add water to volume, and mix. This solution contains 5.76 g of Na per 1000 mL.
for 30 minutes and accurately weighed, to a 500-mL volumetric flask, add water to volume, and mix. This solution contains 5.76 g of Na per 1000 mL.
Potassium stock solution—
Transfer 745.5 mg of potassium chloride, previously dried at 125 for 30 minutes and accurately weighed, to a 1000-mL volumetric flask, add water to volume, and mix. This solution contains 391 mg of K per 1000 mL.
for 30 minutes and accurately weighed, to a 1000-mL volumetric flask, add water to volume, and mix. This solution contains 391 mg of K per 1000 mL.
Surfactant solution—
Transfer 5.0 g of a suitable nonionic surfactant to a 250-mL beaker, add 200 mL of water, and stir to dissolve. Transfer this solution to a 500-mL volumetric flask, dilute with water to volume, and mix. [note—To prevent foaming when using this solution, gently run the solution down the sides of the vessel, and use gentle action when mixing. ]
Diluted sodium solution—
Transfer 50.0 mL of Sodium stock solution and 10.0 mL of Surfactant solution to a 100-mL volumetric flask, dilute with water to volume, and mix gently to prevent foaming.
Standard preparations—
To three separate 500-mL volumetric flasks transfer, respectively, 3.0-, 4.0-, and 5.0-mL portions of Potassium stock solutions. To each flask add 50.0 mL of Sodium stock solution and 10.0 mL of Surfactant solution, dilute with water to volume, and mix gently to prevent foaming. Each mL of these solutions contains 2.346, 3.128, and 3.910 µg of K, respectively.
Assay preparation—
Transfer about 1.4 g of Polacrilin Potassium, previously dried and accurately weighed, to a 50-mL silica crucible, moisten with 4 mL of sulfuric acid, heat over a small flame until the acid has fumed off, moisten the residue with a few drops of sulfuric acid, and ignite strongly. Allow to cool, transfer, with the aid of water, to a 1000-mL volumetric flask, dilute with water to volume, and mix. Transfer 1.00 mL of this solution to a 100-mL volumetric flask, add 20.0 mL of Diluted sodium solution, dilute with water to volume, and mix gently to prevent foaming.
Procedure—
Concomitantly determine the emittances of the Standard preparations and the Assay preparation at 766 nm, with a flame photometer, adjusting the instrument so that the most concentrated Standard preparation gives a reading near 100%. Prepare a standard curve by plotting the standard readings versus the square root of the potassium concentrations. From the curve, determine the concentration, CU, in µg per mL, of potassium in the Assay preparation. Calculate the weight, in mg, of potassium in the portion of Polacrilin Potassium taken by the formula:
100CU
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1892
Pharmacopeial Forum: Volume No. 31(6) Page 1671