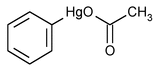
Phenylmercuric Acetate
(fen'' il mer kure' ik as' e tate).
» Phenylmercuric Acetate contains not less than 98.0 percent and not more than 100.5 percent of C8H8HgO2.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
A:
Add 0.5 mL of nitric acid to 0.1 g of it, warm gently until a dark brown color is produced, and dilute with water to 10 mL: the characteristic odor of nitrobenzene is evolved.
B:
To 0.1 g of it add 0.5 mL of sulfuric acid and 1 mL of alcohol, and warm: the characteristic odor of ethyl acetate is evolved.
C:
To 5 mL of a saturated solution in water add a few drops of sodium sulfide TS: a white precipitate is formed, which turns black when the mixture is boiled and then allowed to stand.
Melting range  741
741 :
between 149
:
between 149 and 153
and 153 .
.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Mercuric salts and Heavy metals—
Heat about 100 mg with 15 mL of water, cool, and filter. To the filtrate add a few drops of sodium sulfide TS: the resulting precipitate shows no immediate color.
Polymercurated benzene compounds—
Shake 2.0 g with 100 mL of acetone, and filter. Wash the residue with successive portions of acetone until a total of 50 mL is used, then dry the residue at 105 for 1 hour, and weigh: the weight of the residue does not exceed 30 mg (1.5%).
for 1 hour, and weigh: the weight of the residue does not exceed 30 mg (1.5%).
Assay—
Transfer about 500 mg of Phenylmercuric Acetate, accurately weighed, to a 100-mL flask, add 15 mL of water, 5 mL of formic acid, and 1 g of zinc dust, and reflux for 30 minutes. Cool, filter, and wash the filter paper and the amalgam with water until the washings are no longer acid to litmus. Dissolve the amalgam in 40 mL of 8 N nitric acid. Heat on a steam bath for 3 minutes, and then add 500 mg of urea and enough potassium permanganate TS to produce a permanent pink color. Cool, decolorize the solution with hydrogen peroxide TS, add 1 mL of ferric ammonium sulfate TS, and titrate with 0.1 N ammonium thiocyanate VS. Each mL of 0.1 N ammonium thiocyanate is equivalent to 16.84 mg of C8H8HgO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
USP35–NF30 Page 1890