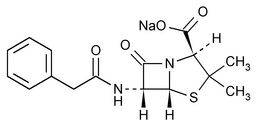
Penicillin G Sodium
(pen'' i sil' in soe' dee um).
4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-6-[(phenylacetyl)amino-], 2S-(2
Monosodium (2S,5R,6R)-3,3-dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate
» Penicillin G Sodium has a potency of not less than 1500 Penicillin G Units and not more than 1750 Penicillin G Units per mg.
Packaging and storage—
Preserve in tight containers.
Labeling—
Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected to further processing during the preparation of injectable dosage forms.
USP Reference standards  11
11 —
—
USP Endotoxin RS
Crystallinity  695
695 :
meets the requirements.
:
meets the requirements.
pH  791
791 :
between 5.0 and 7.5, in a solution containing 60 mg per mL.
:
between 5.0 and 7.5, in a solution containing 60 mg per mL.
Loss on drying  731
731 —
Dry about 100 mg, accurately weighed, in a capillary-stoppered bottle in vacuum at a pressure not exceeding 5 mm of mercury at 60
—
Dry about 100 mg, accurately weighed, in a capillary-stoppered bottle in vacuum at a pressure not exceeding 5 mm of mercury at 60 for 3 hours: it loses not more than 1.5% of its weight.
for 3 hours: it loses not more than 1.5% of its weight.
Other requirements—
Where the label states that Penicillin G Sodium is sterile, it meets the requirements for Sterility and Bacterial endotoxins under Penicillin G Sodium for Injection. Where the label states that Penicillin G Sodium must be subjected to further processing during the preparation of injectable dosage forms, it meets the requirements for Bacterial endotoxins under Penicillin G Sodium for Injection.
Assay—
Mobile phase, Resolution solution, Standard preparation, and Chromatographic system—
Proceed as directed in the Assay under Penicillin G Potassium.
Assay preparation—
Transfer about 5 mg of Penicillin G Sodium, accurately weighed, to a 50-mL volumetric flask, add about 45 mL of water, and shake to dissolve. Dilute with water to volume, and mix.
Procedure—
Proceed as directed in the Assay under Penicillin G Potassium. Calculate the potency, in Penicillin G Units per mg, of Penicillin G Sodium taken by the formula:
(PWS / WU)(rU / rS)
in which WU is the weight of Penicillin G Sodium taken to prepare the Assay preparation, and the other terms are as defined therein.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ahalya Wise, M.S.
Senior Scientific Liaison 1-301-816-8161 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4251
Pharmacopeial Forum: Volume No. 27(1) Page 1814