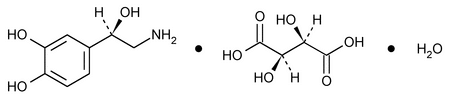Norepinephrine Bitartrate
C8H11NO2·C4H6O6 319.27
1,2-Benzenediol, 4-(2-amino-1-hydroxyethyl)-, (R)-,[R-(R*,R*)]-2,3-dihydroxybutanedioate (1:1) (salt), monohydrate;
(–)- -(Aminomethyl)-3,4-dihydroxybenzyl alcohol tartrate (1:1) (salt), monohydrate
-(Aminomethyl)-3,4-dihydroxybenzyl alcohol tartrate (1:1) (salt), monohydrate


 [69815-49-2].
[69815-49-2].
Anhydrous

 [51-40-1].
[51-40-1].
(nor'' ep i nef' rin bye tar' trate).
C8H11NO2·C4H6O6 319.27
1,2-Benzenediol, 4-(2-amino-1-hydroxyethyl)-, (R)-,[R-(R*,R*)]-2,3-dihydroxybutanedioate (1:1) (salt), monohydrate;
(–)-
Anhydrous
DEFINITION
Norepinephrine Bitartrate contains NLT 97.0% and NMT 102.0% of C8H11NO3·C4H6O6, calculated on the anhydrous basis.
IDENTIFICATION
• B. Procedure
Sample solution:
5 mg/mL in water
Analysis:
Add 1 drop of ferric chloride TS.
Acceptance criteria:
An intensely green color develops.
• C. Procedure
Sample solution:
0.01 µg/mL
Analysis:
Add 1.0 mL of 0.10 N iodine to 10 mL of the Sample solution. Allow to stand for 5 min, and add 2.0 mL of 0.10 N sodium thiosulfate.
Acceptance criteria:
The solution is colorless, or has at most a slight pink or slight violet color (epinephrine and isoproterenol at the same pH, about 3.5, give a strong red-brown or violet color).
ASSAY
• Procedure
Sample solution:
25 mg/mL of Norepinephrine Bitartrate in glacial acetic acid. If necessary warm slightly to effect solution.
Analysis:
Add 2 drops of crystal violet TS to the Sample solution, and titrate with 0.1 N perchloric acid VS. Perform a blank determination, and make any necessary correction (see Titrimetry  541
541 ). Each mL of 0.1 N perchloric acid is equivalent to 31.93 mg of C8H11NO3·C4H6O6.
). Each mL of 0.1 N perchloric acid is equivalent to 31.93 mg of C8H11NO3·C4H6O6.
Acceptance criteria:
97.0%–102.0% on the anhydrous basis
IMPURITIES
Inorganic Impurities
• Residue on Ignition  281
281 :
NMT 0.1%, from 200 mg
:
NMT 0.1%, from 200 mg
Organic Impurities
• Procedure: Limit of Arterenone
Sample solution:
2 mg/mL in water
Analysis:
Determine the absorptivity of the Sample solution (see Spectrophotometry and Light-Scattering  851
851 ) at 310 nm.
) at 310 nm.
Acceptance criteria:
NMT 0.2
SPECIFIC TESTS
• Water Determination, Method I  921
921 :
4.5%–5.8%
:
4.5%–5.8%
ADDITIONAL REQUIREMENTS
• Packaging and Storage:
Preserve in tight, light-resistant containers. Store at controlled room temperature.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 4073
Pharmacopeial Forum: Volume No. 35(5) Page 1165