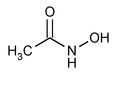
Acetohydroxamic Acid
(a seet'' oh hye'' drox am' ik as' id).
» Acetohydroxamic Acid, dried over phosphorus pentoxide for 16 hours, contains not less than 98.0 percent and not more than 101.0 percent of C2H5NO2.
Packaging and storage—
Preserve in tight containers, and store in a cool, dry place.
Completeness of solution  641
641 —
A 1.0-g portion dissolves in 10 mL of water to yield a clear solution.
—
A 1.0-g portion dissolves in 10 mL of water to yield a clear solution.
Color of solution—
Dissolve 1.0 g in 5 mL of water: the absorbance, determined in a 1-cm cell in the wavelength range between 400 nm and 750 nm in a suitable spectrophotometer, water being used as the blank, is not greater than 0.050.
Identification—
B:
To 10 mL of a solution (1 in 50) add 2 drops of potassium permanganate TS: the pink color of the permanganate disappears.
Loss on drying  731
731 —
Dry it over phosphorus pentoxide for 16 hours: it loses not more than 1.0% of its weight.
—
Dry it over phosphorus pentoxide for 16 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method I  231
231 —
Dissolve 1 g in 23 mL of water, and add 2 mL of 1 N acetic acid: the limit is 0.002%.
—
Dissolve 1 g in 23 mL of water, and add 2 mL of 1 N acetic acid: the limit is 0.002%.
Limit of hydroxylamine—
Phosphate buffer—
Dissolve 1.36 g of monobasic potassium phosphate in about 950 mL of water, adjust with 1 M potassium hydroxide to a pH of 7.4, dilute with water to 1000 mL, and mix.
Pyridoxal 5-phosphate solution—
Dissolve 50 mg of pyridoxal 5-phosphate monohydrate in 50 mL of Phosphate buffer in a low-actinic flask. Prepare fresh before use.
Standard solutions—
Dissolve an accurately weighed quantity of hydroxylamine hydrochloride in water to obtain a final concentration of 2.0 mg per mL. To separate 100-mL volumetric flasks, transfer 5.0, 10.0, and 15.0 mL of the hydroxylamine stock solution, respectively, dilute each flask with water to volume, and mix.
Test solution—
Transfer an accurately weighed quantity of about 1500 mg of Acetohydroxamic Acid, previously dried, to a 100-mL beaker, and dissolve in a sufficient amount of water to cover the electrode of a calibrated pH meter (about 60 mL). While stirring, adjust with 0.05 M potassium hydroxide to a pH of 7.4. Quantitatively transfer the contents of the beaker, with the aid of small portions of water, to a 100-mL volumetric flask, dilute with water to volume, and mix.
Procedure—
Transfer 2.0 mL of each Standard solution and the Test solution into separate 100-mL volumetric flasks. Pipet 2.0 mL of water into a 100-mL volumetric flask for the reagent blank. To each flask, add 4.0 mL of Pyridoxal 5-phosphate solution, and mix. After 8 minutes, accurately timed, dilute the contents of each flask with Phosphate buffer to volume. Immediately determine the fluorescence intensities of the solutions from the Standard solutions and the Test solution in a fluorometer at an excitation wavelength of 350 nm and an emission wavelength of 450 nm, setting the instrument to zero with the reagent blank. Determine the best-fit straight line from the fluorescence intensities of the three Standard solutions versus the hydroxylamine hydrochloride concentrations, in µg per mL. From the best-fit straight line, determine the concentration, in µg per mL, of hydroxylamine hydrochloride in the Test solution. Calculate the percentage of hydroxylamine in the portion of Acetohydroxamic Acid taken by the formula:
(33.03/69.50)(10C/W)
in which 33.03 and 69.50 are the molecular weights of hydroxylamine and hydroxylamine hydrochloride, respectively; C is the concentration, in µg per mL, of hydroxylamine hydrochloride in the Test solution; and W is the weight, in mg, of Acetohydroxamic Acid taken. Not more than 0.5% is found.
Assay—
Ferric chloride solution—
Dissolve 4 g of ferric chloride in about 50 mL of 0.1 N hydrochloric acid, dilute with the same solvent to make 200 mL, and mix.
Standard preparation—
Dissolve an accurately weighed quantity of USP Acetohydroxamic Acid RS in 0.1 N hydrochloric acid to obtain a solution having a known concentration of about 500 µg per mL.
Assay preparation—
Transfer about 250 mg of Acetohydroxamic Acid, previously dried and accurately weighed, to a 500-mL volumetric flask, dilute with 0.1 N hydrochloric acid to volume, and mix.
Procedure—
Transfer 10.0 mL each of the Standard preparation, the Assay preparation, and 0.1 N hydrochloric acid to provide the blank, to separate 100-mL volumetric flasks. To each flask add about 50 mL of 0.1 N hydrochloric acid and 10.0 mL of Ferric chloride solution, mix, dilute with 0.1 N hydrochloric acid to volume, and mix. Without delay, concomitantly determine the absorbances of the solutions from the Standard preparation and the Assay preparation at the wavelength of maximum absorbance at about 502 nm, with a suitable spectrophotometer, using the blank to set the instrument. Calculate the quantity, in mg, of C2H5NO2 in the portion of Acetohydroxamic Acid taken by the formula:
0.5C(AU / AS)
in which C is the concentration, in µg per mL, of USP Acetohydroxamic Acid RS in the Standard preparation; and AU and AS are the absorbances of the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2066
Pharmacopeial Forum: Volume No. 30(5) Page 1579