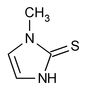Methimazole
(meth im' a zole).
» Methimazole contains not less than 98.0 percent and not more than 101.0 percent of C4H6N2S, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Identification—
B:
Mercuric chloride TS produces in a solution (1 in 200) a white precipitate, but no precipitation is produced by trinitrophenol TS. The solution is colored intensely blue by molybdophosphotungstate TS.
Melting range  741
741 :
between 143
:
between 143 and 146
and 146 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 2 hours: it loses not more than 0.5% of its weight.
for 2 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Selenium  291
291 :
0.003%, a 200-mg specimen being used.
:
0.003%, a 200-mg specimen being used.
Ordinary impurities  466
466 —
—
Test solution:
ethyl acetate.
Standard solution:
ethyl acetate.
Eluant:
a mixture of toluene, isopropyl alcohol, and ammonium hydroxide (70:29:1), in a nonequilibrated chamber.
Visualization:
2.
Assay—
Dissolve about 250 mg of Methimazole, accurately weighed, in 75 mL of water. Add from a buret 15 mL of 0.1 N sodium hydroxide VS, mix, and add, with agitation, about 30 mL of 0.1 N silver nitrate. Add 1 mL of bromothymol blue TS, and continue the titration with the 0.1 N sodium hydroxide VS until a permanent, blue-green color is produced. Each mL of 0.1 N sodium hydroxide is equivalent to 11.42 mg of C4H6N2S.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3850