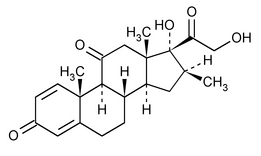
Meprednisone
(me pred' ni sone).
Pregna-1,4-diene-3,11,20-trione, 17,21-dihydroxy-16-methyl-, (16
17,21-Dihydroxy-16
» Meprednisone contains not less than 97.5 percent and not more than 102.5 percent of C22H28O5, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers, and avoid exposure to excessive heat.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
10 µg per mL.
Medium:
methanol.
Absorptivities at 238 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
Thin-Layer Chromatographic Identification Test  201
201 —
—
Test solution:
20 mg per mL, in a mixture of toluene and alcohol (1:1).
Spray reagent:
10% (v/v) sulfuric acid in alcohol solution.
Procedure—
Proceed as directed in the chapter. Locate the spots on the plate by spraying with Spray reagent and heating at 105 for 10 minutes.
for 10 minutes.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 1.0% of its weight.
for 3 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Assay—
Standard preparation—
Prepare as directed under Single-Steroid Assay  511
511 , using USP Meprednisone RS.
, using USP Meprednisone RS.
Assay preparation—
Accurately weigh about 20 mg of Meprednisone, previously dried, dissolve it in a sufficient quantity of a mixture of alcohol and chloroform (1:1) to make 10.0 mL, and mix.
Procedure—
Proceed as directed for Procedure under Single-Steroid Assay  511
511 , using a solvent system consisting of chloroform, methanol, and water (180:15:1), through the fourth sentence of the second paragraph under Procedure. Then centrifuge the tubes for 5 minutes, and determine the absorbances of the supernatants in 1-cm cells at the wavelength of maximum absorbance at about 238 nm, with a suitable spectrophotometer, against the blank. Calculate the quantity, in mg, of C22H28O5 in the portion of Meprednisone taken by the formula:
, using a solvent system consisting of chloroform, methanol, and water (180:15:1), through the fourth sentence of the second paragraph under Procedure. Then centrifuge the tubes for 5 minutes, and determine the absorbances of the supernatants in 1-cm cells at the wavelength of maximum absorbance at about 238 nm, with a suitable spectrophotometer, against the blank. Calculate the quantity, in mg, of C22H28O5 in the portion of Meprednisone taken by the formula:
10C(AU / AS)
in which C is the concentration, in mg per mL, of USP Meprednisone RS in the Standard preparation; and AU and AS are the absorbances of the solutions from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Mary S. Waddell
Scientific Liaison 1-301-816-8124 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3807
Pharmacopeial Forum: Volume No. 27(2) Page 2174