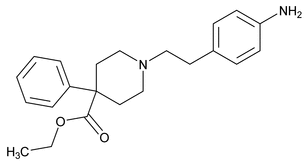
Anileridine
(an'' i ler' i deen).
4-Piperidinecarboxylic acid, 1-[2-(4-aminophenyl)ethyl]-4-phenyl-, ethyl ester.
Ethyl 1-(p-aminophenethyl)-4-phenylisonipecotate
» Anileridine contains not less than 98.5 percent and not more than 101.0 percent of C22H28N2O2, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight, light-resistant containers. Store at room temperature.
Identification—
A:
Dissolve 40 mg in 2.3 mL of 0.1 N hydrochloric acid in a 100-mL volumetric flask, dilute with water to volume, and mix (Stock solution). Transfer 4.0 mL of this solution to a 100-mL volumetric flask, and add 25 mL of pH 7.0 Buffer solution. (Prepare the Buffer solution by dissolving 22.73 g of anhydrous dibasic sodium phosphate and 14.52 g of monobasic potassium phosphate in water to make 1000 mL. Dilute 25 mL of the buffer with water to 100 mL: the pH, determined potentiometrically, is 7.0 ± 0.05.) Dilute with water to volume, and mix (Solution A). Transfer 20.0 mL of the Stock solution to a 100-mL volumetric flask, add 25 mL of the pH 7.0 Buffer solution, dilute with water to volume, and mix (Solution B): the UV absorption spectrum of Solution A exhibits a maximum at 234 ± 1 nm; and the UV absorption spectrum of Solution B exhibits a maximum at 285 ± 2 nm. The ratio 5A234 / A285 is about 8.8.
B:
To 5 mL of a solution in 0.1 N hydrochloric acid (1 in 5000), add 2 mL of a solution of p-dimethylaminobenzaldehyde in alcohol (1 in 100): a yellow color develops immediately.
Water, Method I  921
921 :
not more than 1.0%.
:
not more than 1.0%.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chloride  221
221 —
Dissolve 180 mg in a mixture of 1 mL of nitric acid and 40 mL of water: the solution shows no more chloride than corresponds to 0.10 mL of 0.020 N hydrochloric acid (0.040%).
—
Dissolve 180 mg in a mixture of 1 mL of nitric acid and 40 mL of water: the solution shows no more chloride than corresponds to 0.10 mL of 0.020 N hydrochloric acid (0.040%).
Assay—
Dissolve about 350 mg of Anileridine, accurately weighed, in 50 mL of glacial acetic acid, add 1 drop of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a blue-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 17.62 mg of C22H28N2O2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Clydewyn M. Anthony, Ph.D.
Senior Scientific Liaison 1-301-816-8139 |
(SM22010) Monographs - Small Molecules 2 |
USP35–NF30 Page 2219
Pharmacopeial Forum: Volume No. 29(6) Page 1846