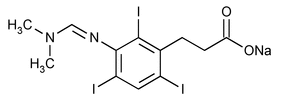
Ipodate Sodium
(eye' poe date soe' dee um).
Benzenepropanoic acid, 3-[[(dimethylamino)methylene]amino]-2,4,6-triiodo-, sodium salt.
Sodium 3-[[(dimethylamino)methylene]amino]-2,4,6-triiodohydrocinnamate
» Ipodate Sodium contains not less than 97.5 percent and not more than 102.5 percent of C12H12I3N2NaO2, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
10 µg per mL.
Medium:
methanol.
Absorptivities at 235 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
Heat about 500 mg in a porcelain crucible over a free flame: violet vapors of iodine are evolved.
D:
It responds to the flame test for Sodium  191
191 .
.
Loss on drying  731
731 —
Dry it in vacuum at 60
—
Dry it in vacuum at 60 for 3 hours: it loses not more than 0.5% of its weight.
for 3 hours: it loses not more than 0.5% of its weight.
Iodide or iodine—
Dissolve about 200 mg in 10 mL of 6 N acetic acid, add 2 mL of 1 N sulfuric acid and 15 mL of chloroform, and shake vigorously. Allow the layers to separate: the chloroform layer shows not more than a faint violet color. Add 1 mL of 0.1 N potassium iodate, shake vigorously, and allow the layers to separate: the chloroform layer shows at most a slight trace of violet color.
Heavy metals, Method II  231
231 :
0.003%.
:
0.003%.
Assay—
Transfer about 300 mg of Ipodate Sodium, accurately weighed, to a 250-mL flask, add 30 mL of 1.25 N sodium hydroxide and 0.5 g of powdered zinc, and reflux the mixture for 60 minutes. Cool, wash the condenser with 20 mL of water, and filter the mixture. Wash the flask and the filter with small portions of water, adding the washings to the filtrate. Add to the filtrate 5 mL of glacial acetic acid and 3 mL of a mixture of 2 drops of nitric acid in 5 mL of water, then add 3 drops of eosin Y TS, and titrate with 0.05 N silver nitrate VS until the entire mixture changes to a permanent pink color. Each mL of 0.05 N silver nitrate is equivalent to 10.33 mg of C12H12I3N2NaO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3554