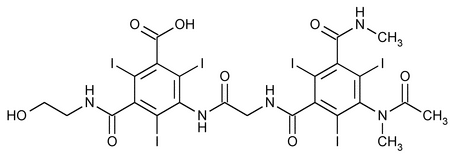
Ioxaglic Acid
(eye'' ox ag' lik as' id).
Benzoic acid, 3-[[[[3-(acetylmethylamino)-2,4,6-triiodo-5-[(methylamino)carbonyl]benzoyl]amino]acetyl]amino]-5-[[(2-hydroxyethyl)amino]carbonyl]-2,4,6-triiodo-.
N-(2-Hydroxyethyl)-2,4,6-triiodo-5[2-[2,4,6-triiodo-3-(N-methylacetamido)-5-(methylcarbamoyl)benzamido]acetamido]isophthalamic acid
» Ioxaglic Acid contains not less than 98.5 percent and not more than 101.5 percent of C24H21I6N5O8, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification—
A: Thin-Layer Chromatographic Identification Test  201
201 —
—
Test solution—
Dissolve 50 mg in 5 mL of methanol.
Application volume:
5 µL.
Developing solvent system:
butyl alcohol, water, and acetic acid (50:25:11).
Procedure—
Proceed as directed in the chapter. The RF values of the two spots obtained from the Test solution correspond to those obtained from the Standard solution.
B:
Heat about 500 mg in a crucible: violet vapors are evolved.
Water, Method I  921
921 :
not more than 5%.
:
not more than 5%.
Residue on ignition  281
281 :
not more than 0.1%, the residue being moistened with 2 mL of 35% sulfuric acid and ignited at 600
:
not more than 0.1%, the residue being moistened with 2 mL of 35% sulfuric acid and ignited at 600 .
.
Heavy metals—
Test solution—
Dissolve 2 g of Ioxaglic Acid in 16 mL of 0.1 N sodium hydroxide in a glass-stoppered 25-mL graduated cylinder, dilute with water to 20 mL, and mix. Transfer 15 mL of this solution to a 50-mL color-comparison tube.
Standard solution—
Transfer 2.0 mL of Standard Lead Solution (20 µg of Pb) (see Heavy Metals  231
231 ) to a 50-mL color-comparison tube, add 5 mL of the Test solution and 15 mL of water, and mix.
) to a 50-mL color-comparison tube, add 5 mL of the Test solution and 15 mL of water, and mix.
Procedure—
Adjust the Test solution and the Standard solution with 1 N acetic acid to a pH of between 3 and 4. Add 1 mL of a solution of sodium sulfide containing 1.16 mg per mL, mix, allow to stand for 5 minutes, and view downward over a white surface: the color of the solution from the Test solution is not darker than that of the solution from the Standard solution (0.002%).
Free iodine and iodide—
Test solution—
Dissolve 2 g of Ioxaglic Acid in 20 mL of 0.1 N sodium hydroxide in a 50-mL centrifuge tube. Add 15 mL of 2 N sulfuric acid, mix on a vortex mixer, centrifuge for 15 minutes, and decant the supernatant layer into a glass-stoppered 100-mL graduated cylinder. Repeat the sulfuric acid washing and centrifugation twice more, decanting each supernatant layer into the 100-mL graduated cylinder.
Procedure—
Add 5 mL of toluene, shake vigorously, and allow the layers to separate: the toluene layer shows no red color. Add 1 mL of sodium nitrite solution (1 in 50), shake, and allow the layers to separate: any red color in the toluene layer is not darker than that obtained when a mixture of 2.0 mL of potassium iodide solution (1 in 4000), 25 mL of water, and 15 mL of 2 N sulfuric acid is substituted for the Test solution (0.02% of iodide).
Assay—
Transfer about 500 mg of Ioxaglic Acid, accurately weighed, to a glass-stoppered 125-mL conical flask, and add 12 mL of 5 N sodium hydroxide, 20 mL of water, and 1 g of powdered zinc. Connect the flask to a reflux condenser, and reflux for 30 minutes. Cool the flask to room temperature, rinse the condenser with 20 mL of water, disconnect the flask from the condenser, and filter the mixture. Rinse the flask and the filter thoroughly, adding the rinsing to the filtrate. Add 40 mL of 2 N sulfuric acid, and titrate immediately with 0.05 N silver nitrate VS, determining the endpoint potentiometrically, using silver-calomel electrodes and an agar–potassium nitrate salt bridge. Each mL of 0.05 N silver nitrate is equivalent to 10.57 mg of C24H21I6N5O8.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3548
Pharmacopeial Forum: Volume No. 29(6) Page 1911