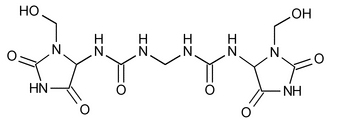
Imidurea
(im id'' ue ree' a; im id'' ure ee' a).
N,N¢¢-Methylenebis[N¢-[3-(hydroxymethyl)-2,5-dioxo-4-imidazolidinyl]urea].
1,1¢-Methylenebis[3-[3-(hydroxymethyl)-2,5-dioxo-4-imidazolidinyl]urea].
» Imidurea contains not less than 26.0 percent and not more than 28.0 percent of nitrogen (N), calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Color and clarity of solution—
Dissolve 3.0 g in 7.0 mL of water in a test tube: the solution is clear and colorless.
Identification, Infrared Absorption  197K
197K .
.
pH  791
791 :
between 6.0 and 7.5, in a solution (1 in 100).
:
between 6.0 and 7.5, in a solution (1 in 100).
Loss on drying  731
731 —
Dry in vacuum over phosphorus pentoxide for 48 hours: it loses not more than 3.0% of its weight.
—
Dry in vacuum over phosphorus pentoxide for 48 hours: it loses not more than 3.0% of its weight.
Residue on ignition  281
281 :
not more than 3.0%.
:
not more than 3.0%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Nitrogen content—
Place about 150 mg of Imidurea, accurately weighed, in a 500-mL Kjeldahl flask, and add 8.0 g of anhydrous sodium sulfate, 0.5 g of anhydrous cupric sulfate, 0.1 g of yellow mercuric oxide, and 11 mL of sulfuric acid. Incline the flask at an angle of about 45 , and gently heat the mixture, keeping the temperature below the boiling point until frothing has ceased. Increase the heat until the acid boils briskly, and continue the heating until the solution has become clear green in color or practically colorless for 30 minutes. Allow to cool, cautiously add 225 mL of water, mix the contents of the flask, and again cool. Add cautiously 1.0 g of zinc metal dust, 2.0 g of sodium thiosulfate, and 18.0 g of sodium hydroxide pellets, and without delay connect the flask to a Kjeldahl connecting bulb (trap), previously attached to a condenser, the delivery tube of which dips beneath the surface of 50 mL of boric acid solution (1 in 25) contained in a 300-mL conical flask. Mix the contents of the Kjeldahl flask by gentle rotation, and distill about 125 mL into the receiver. Add not less than 3 drops of methyl red-methylene blue TS to the contents of the receiving vessel, and determine the ammonia by titration with 0.1 N hydrochloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N hydrochloric acid is equivalent to 1.401 mg of nitrogen.
, and gently heat the mixture, keeping the temperature below the boiling point until frothing has ceased. Increase the heat until the acid boils briskly, and continue the heating until the solution has become clear green in color or practically colorless for 30 minutes. Allow to cool, cautiously add 225 mL of water, mix the contents of the flask, and again cool. Add cautiously 1.0 g of zinc metal dust, 2.0 g of sodium thiosulfate, and 18.0 g of sodium hydroxide pellets, and without delay connect the flask to a Kjeldahl connecting bulb (trap), previously attached to a condenser, the delivery tube of which dips beneath the surface of 50 mL of boric acid solution (1 in 25) contained in a 300-mL conical flask. Mix the contents of the Kjeldahl flask by gentle rotation, and distill about 125 mL into the receiver. Add not less than 3 drops of methyl red-methylene blue TS to the contents of the receiving vessel, and determine the ammonia by titration with 0.1 N hydrochloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N hydrochloric acid is equivalent to 1.401 mg of nitrogen.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1827