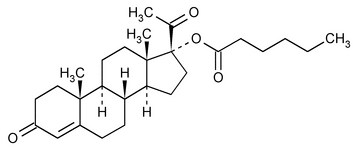
Hydroxyprogesterone Caproate
(hye drox'' ee proe jes' ter one kap' roe ate).
Pregn-4-ene-3,20-dione, 17-[(1-oxohexyl)oxy]-.
17-Hydroxypregn-4-ene-3,20-dione hexanoate
» Hydroxyprogesterone Caproate contains not less than 97.0 percent and not more than 103.0 percent of C27H40O4, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification, Infrared Absorption  197K
197K .
.
Melting range  741
741 :
between 120
:
between 120 and 124
and 124 .
.
Water, Method I  921
921 :
not more than 0.1%.
:
not more than 0.1%.
Free n-caproic acid—
Dissolve 0.20 g in 25 mL of alcohol that previously has been neutralized to a faint pink color following the addition of 2 or 3 drops of phenolphthalein TS. Promptly titrate with 0.020 N sodium hydroxide: not more than 0.50 mL of 0.020 N sodium hydroxide is required (0.58%).
Ordinary impurities  466
466 —
—
Test solution:
chloroform.
Standard solution:
chloroform.
Eluant:
a mixture of chloroform and ethyl acetate (3:1).
Visualization:
5; then view under long-wavelength UV light.
Assay—
Transfer about 50 mg of Hydroxyprogesterone Caproate, accurately weighed, to a 100-mL volumetric flask, add alcohol to volume, and mix. Dilute 2.0 mL of this solution with alcohol to volume in a second 100-mL volumetric flask, and mix. Dissolve in alcohol a suitable quantity of USP Hydroxyprogesterone Caproate RS, and dilute quantitatively and stepwise with alcohol to obtain a Standard solution having a known concentration of about 10 µg per mL. Concomitantly determine the absorbances of both solutions in 1-cm cells at the wavelength of maximum absorbance at about 240 nm, using alcohol as the blank. Calculate the quantity, in mg, of C27H40O4 in the Hydroxyprogesterone Caproate taken by the formula:
5C(AU / AS)
in which C is the concentration, in µg per mL, of USP Hydroxyprogesterone Caproate RS in the Standard solution; and AU and AS are the absorbances of the solution of Hydroxyprogesterone Caproate and the Standard solution, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3455
Pharmacopeial Forum: Volume No. 29(5) Page 1506