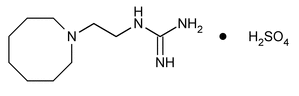Guanethidine Monosulfate
(gwahn eth' i deen mon'' oh sul' fate).
Guanidine, [2-(hexahydro-1(2H)-azocinyl)ethyl]-, sulfate (1:1).
[2-(Hexahydro-1(2H)-azocinyl)ethyl]guanidine sulfate (1:1)
» Guanethidine Monosulfate contains not less than 97.0 percent and not more than 103.0 percent of C10H22N4·H2SO4, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
B:
Dissolve 2.5 mg in 10 mL of water. Add 2 mL of a solution prepared by dissolving 500 mg of 1-naphthol, 3 g of sodium hydroxide, and 8 g of sodium carbonate in water to make 50 mL, and 1 mL of a solution of 2,3-butanedione (1 in 2000). Allow to stand at room temperature: an intense, pinkish-red color develops.
pH  791
791 :
between 4.7 and 5.7, in a solution containing 20 mg per mL.
:
between 4.7 and 5.7, in a solution containing 20 mg per mL.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 to constant weight: it loses not more than 0.5% of its weight.
to constant weight: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Assay—
Sodium nitroferricyanide–potassium ferricyanide solution—
Dissolve 1 g of sodium nitroferricyanide and 1 g of potassium ferricyanide in water to make 100 mL, and mix.
Standard preparation—
Dissolve a suitable quantity of USP Guanethidine Monosulfate RS, accurately weighed, in 1 N sulfuric acid to obtain a solution having a known concentration of about 1 mg per mL.
Assay preparation—
Transfer about 50 mg of Guanethidine Monosulfate, accurately weighed, to a 50-mL volumetric flask, dissolve in 1 N sulfuric acid, add 1 N sulfuric acid to volume, and mix.
Procedure—
Pipet 2 mL each of the Assay preparation and the Standard preparation, and 2 mL of 1 N sulfuric acid to provide the blank, into separate glass-stoppered, 40-mL centrifuge tubes. Add 10.0 mL of water to each tube, and mix. To each tube add 10.0 mL of Sodium nitroferricyanide–potassium ferricyanide solution, mix, add 4.0 mL of 1 N sodium hydroxide, mix, and allow to stand for 20 minutes, accurately timed. Concomitantly determine the absorbances of both solutions in 1-cm cells against the blank at the wavelength of maximum absorbance at about 500 nm, with a suitable spectrophotometer. Calculate the quantity, in mg, of C10H22N4·H2SO4 in the Guanethidine Monosulfate taken by the formula:
50C(AU / AS)
in which C is the concentration, in mg per mL, of USP Guanethidine Monosulfate RS in the Standard preparation; and AU and AS are the absorbances of the solutions from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3389