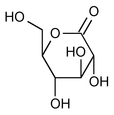
Gluconolactone
(gloo'' kon oh lak' tone).
» Gluconolactone contains not less than 99.0 percent and not more than 101.0 percent of C6H10O6.
Packaging and storage—
Preserve in well-closed containers.
Identification—
Dissolve about 0.5 g in 5 mL of warm water in a test tube. Add 1 mL of freshly distilled phenylhydrazine, and heat on a steam bath for 30 minutes. Cool the solution, induce crystallization by scratching the inner surface of the test tube with a glass rod, and collect the crystals of the phenylhydrazide of gluconic acid. Dissolve the crystals in 10 mL of hot water to which a small amount of activated charcoal has been added, filter, recrystallize, wash the crystals with 2 mL of cold water, and dry the crystals at 105 for 1 hour: the crystals so obtained melt between 195
for 1 hour: the crystals so obtained melt between 195 and 200
and 200 .
.
Lead  251
251 —
Prepare a Test Preparation as directed in the chapter, and use 10 mL of Diluted Standard Lead Solution (10 µg of lead) for the test: the limit is 0.001%.
—
Prepare a Test Preparation as directed in the chapter, and use 10 mL of Diluted Standard Lead Solution (10 µg of lead) for the test: the limit is 0.001%.
Heavy metals  231
231 —
Dissolve 1 g in 25 mL of water: the limit is 0.002%.
—
Dissolve 1 g in 25 mL of water: the limit is 0.002%.
Reducing substances—
Transfer 10.0 g to a 400-mL beaker, add 40 mL of water, and swirl to dissolve. Add 2 drops of phenolphthalein TS, and neutralize with sodium hydroxide solution (1 in 2). Dilute with water to about 50 mL, and add 50 mL of alkaline cupric tartrate TS. Heat so that the solution begins to boil in 4 minutes, and allow boiling to continue for 120 seconds. Pass the suspension through a medium-porosity filtering crucible, and wash the filter with three 5-mL portions of water. Place the crucible in an upright position in the original beaker, add 5 mL of water and 3 mL of nitric acid to the crucible, mix with a glass rod to ensure complete solution of the cuprous oxide, and wash the solution from the crucible into a beaker with the aid of 5 mL of water. Add bromine TS, usually 5 to 10 mL, until the solution becomes yellow in color, and dilute with water to about 75 mL. Add a few glass beads, boil until the bromine has been driven off, and cool. Slowly add ammonium hydroxide until a deep blue color appears, then adjust with glacial acetic acid to a pH of 4, and add water to make about 100 mL. Add 4 g of potassium iodide, and titrate with 0.1 N sodium thiosulfate VS, adding starch TS just before the endpoint is reached: not more than 16.1 mL of 0.1 N sodium thiosulfate is consumed.
Assay—
Dissolve about 600 mg of Gluconolactone, accurately weighed, in 100 mL of water in a 300-mL conical flask, add 50.0 mL of 0.1 N sodium hydroxide VS, and allow to stand for 15 minutes. Add phenolphthalein TS, and titrate the excess alkali with 0.1 N hydrochloric acid VS. Perform a blank determination (see Residual Titrations under Titrimetry  541
541 ). Each mL of 0.1 N sodium hydroxide is equivalent to 17.81 mg of C6H10O6.
). Each mL of 0.1 N sodium hydroxide is equivalent to 17.81 mg of C6H10O6.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
USP35–NF30 Page 3346