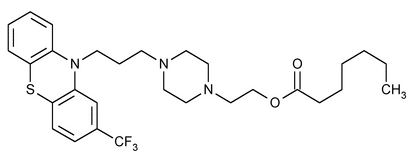
Fluphenazine Enanthate
(floo fen' a zeen e nan' thate).
Heptanoic acid, 2-[4-[3-[2-(trifluoromethyl)-10H-phenothiazin-10-yl]propyl]-1-piperazinyl]ethyl ester.
2-[4-[3-[2-(Trifluoromethyl)phenothiazin-10-yl]propyl]-1-piperazinyl]ethyl heptanoate
» Fluphenazine Enanthate contains not less than 97.0 percent and not more than 103.0 percent of C29H38F3N3O2S, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
USP Reference standards  11
11 —
—
USP Fluphenazine Enanthate Dihydrochloride RS 
C29H38F3N3O2S·2HCl 622.63

C29H38F3N3O2S·2HCl 622.63
[Note—Throughout the following procedures, protect test or assay specimens, the USP Reference Standard, and solutions containing them, by conducting the procedures without delay, under subdued light, or using low-actinic glassware. ]
Identification—
A:
Place about 50 mg of Fluphenazine Enanthate and about 50 mg of USP Fluphenazine Enanthate Dihydrochloride RS in separate, glass-stoppered, small centrifuge tubes, and treat each tube as follows. Add 1.5 mL of sodium hydroxide solution (1 in 250), and mix. Add 2 mL of carbon disulfide, shake vigorously for 2 minutes, and centrifuge. Dry the lower, clear layer by filtering it through 2 g of anhydrous sodium sulfate: the IR absorption spectrum of the test preparation, determined in a 0.1-mm cell, exhibits maxima only at the same wavelengths as that of the Standard preparation, similarly measured.
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
10 µg per mL.
Medium:
methanolic hydrochloric acid (8.5 in 1000).
Absorptivities at 258 nm, calculated on the dried basis, do not differ by more than 2.5%.
Loss on drying  731
731 —
Dry it in vacuum at 60
—
Dry it in vacuum at 60 for 3 hours: it loses not more than 1.0% of its weight.
for 3 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Heavy metals, Method II  231
231 :
0.003%.
:
0.003%.
Ordinary impurities  466
466 —
—
Test solution:
alcohol.
Standard solution:
alcohol.
Eluant:
a mixture of alcohol, glacial acetic acid, and water (3:1:1).
Visualization:
1.
Assay—
Dissolve about 500 mg of Fluphenazine Enanthate, accurately weighed, in 50 mL of glacial acetic acid, add 1 drop of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a blue-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 27.49 mg of C29H38F3N3O2S.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3250
Pharmacopeial Forum: Volume No. 29(6) Page 1887