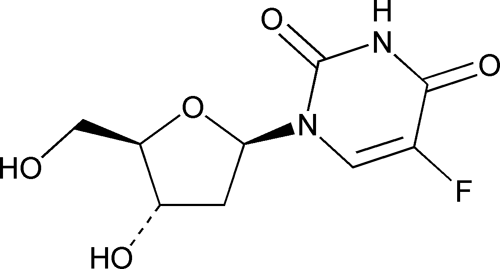
Floxuridine
(flox ure' i deen).
» Floxuridine contains not less than 98.5 percent and not more than 101.0 percent of C9H11FN2O5, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Labeling—
Where it is intended for use in preparing injectable dosage forms, the label states that it is sterile or must be subjected for further processing during the preparation of injectable dosage forms.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
20 µg per mL.
Medium:
0.1 N potassium hydroxide.
Absorptivities at 268 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
Dissolve about 200 mg in 10 mL of water, and add a few drops of bromine TS: the bromine color is discharged.
Melting range, Class I  741
741 :
between 145
:
between 145 and 153
and 153 , but the range between beginning and end of melting does not exceed 2
, but the range between beginning and end of melting does not exceed 2 .
.
Loss on drying  731
731 —
Dry it in vacuum over silica gel at 60
—
Dry it in vacuum over silica gel at 60 for 4 hours: it loses not more than 0.2% of its weight.
for 4 hours: it loses not more than 0.2% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Limit of fluoride ions—
[note—Use plasticware throughout this test. ]
pH 5.25 Buffer—
Dissolve 110 g of sodium chloride and 1 g of sodium citrate in 700 mL of water in a 2000-mL volumetric flask. Cautiously add 150 g of sodium hydroxide, and dissolve with shaking. Cool to room temperature, and, while stirring, cautiously add 450 mL of glacial acetic acid to the cooled solution. Cool, add 600 mL of isopropyl alcohol, dilute with water to volume, and mix: the pH of this solution is between 5.0 and 5.5.
Standard stock solution—
Transfer 221 mg of sodium fluoride, previously dried at 150 for 4 hours, to a 100-mL volumetric flask, add about 20 mL of water, and mix to dissolve. Add 1.0 mL of sodium hydroxide solution (1 in 2500), dilute with water to volume, and mix. Each mL of this solution contains 1 mg of fluoride ions. Store in a tightly closed, plastic container.
for 4 hours, to a 100-mL volumetric flask, add about 20 mL of water, and mix to dissolve. Add 1.0 mL of sodium hydroxide solution (1 in 2500), dilute with water to volume, and mix. Each mL of this solution contains 1 mg of fluoride ions. Store in a tightly closed, plastic container.
Standard solutions—
Dilute portions of the Standard stock solution quantitatively and stepwise with pH 5.25 Buffer to obtain 100-mL solutions having concentrations of 1, 3, 5, and 10 µg per mL.
Test solution—
Transfer 1.0 g of Floxuridine to a 100-mL volumetric flask, dissolve in and dilute with pH 5.25 Buffer to volume, and mix.
Procedure—
Concomitantly measure the potential (see Titrimetry  541
541 ), in mV, of the Standard solutions and the Test solution, with a pH meter capable of a minimum reproducibility of ±0.2 mV, equipped with a glass-sleeved calomel-fluoride specific-ion electrode system. [note—When taking measurements, immerse the electrodes in the solution, which has been transferred to a 150-mL beaker containing a polytef-coated stirring bar. Allow to stir on a magnetic stirrer having an insulated top until equilibrium is attained (1 to 2 minutes), and record the potential. Rinse and dry the electrodes between measurements, taking care to avoid damaging the crystal of the specific-ion electrode. ] Plot the logarithm of the fluoride-ion concentrations, in µg per mL, of the Standard solutions versus potential, in mV. From the measured potential of the Test solution and the standard curve, determine the concentration, in µg per mL, of fluoride ions in the Test solution: not more than 0.05% is found.
), in mV, of the Standard solutions and the Test solution, with a pH meter capable of a minimum reproducibility of ±0.2 mV, equipped with a glass-sleeved calomel-fluoride specific-ion electrode system. [note—When taking measurements, immerse the electrodes in the solution, which has been transferred to a 150-mL beaker containing a polytef-coated stirring bar. Allow to stir on a magnetic stirrer having an insulated top until equilibrium is attained (1 to 2 minutes), and record the potential. Rinse and dry the electrodes between measurements, taking care to avoid damaging the crystal of the specific-ion electrode. ] Plot the logarithm of the fluoride-ion concentrations, in µg per mL, of the Standard solutions versus potential, in mV. From the measured potential of the Test solution and the standard curve, determine the concentration, in µg per mL, of fluoride ions in the Test solution: not more than 0.05% is found.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Other requirements—
Where the label states that Floxuridine is sterile, it meets the requirements for Sterile Solids under Injections  1
1 and for Pyrogen under Floxuridine for Injection. Where the label states that Floxuridine must be subjected to further processing during the preparation of injectable dosage forms, it meets the requirements for Pyrogen under Floxuridine for Injection.
and for Pyrogen under Floxuridine for Injection. Where the label states that Floxuridine must be subjected to further processing during the preparation of injectable dosage forms, it meets the requirements for Pyrogen under Floxuridine for Injection.
Assay—
Dissolve about 800 mg of Floxuridine, accurately weighed, in 80 mL of dimethylformamide, and titrate with 0.1 N tetrabutylammonium hydroxide VS, determining the endpoint potentiometrically using a calomel-glass electrode system. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N tetrabutylammonium hydroxide is equivalent to 24.62 mg of C9H11FN2O5.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientific Liaison 1-301-816-8394 |
(SM12010) Monographs - Small Molecules 1 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3203
Pharmacopeial Forum: Volume No. 29(6) Page 1886