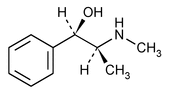
Ephedrine
(e fed' rin).
Benzenemethanol,
(
Hemihydrate 174.24
» Ephedrine is anhydrous or contains not more than one-half molecule of water of hydration. It contains not less than 98.5 percent and not more than 100.5 percent of C10H15NO, calculated on the anhydrous basis.
Packaging and storage—
Preserve in tight, light-resistant containers, in a cold place.
Labeling—
Label it to indicate whether it is hydrous or anhydrous. Where the quantity of Ephedrine is indicated in the labeling of any preparation containing Ephedrine, this shall be understood to be in terms of anhydrous Ephedrine.
Identification—
Accurately weigh about 100 mg, and add by buret the exact volume of 0.1 N sulfuric acid, determined in the Assay, to neutralize it. Dilute with water in a volumetric flask to 25 mL. Mix 2 mL with 10 mL of alcohol, and evaporate on a steam bath with the aid of a current of air to dryness: the residue so obtained responds to Identification test A under Ephedrine Sulfate.
Specific rotation  781S
781S :
between –40.3
:
between –40.3 and –43.3
and –43.3 .
.
Test solution:
25 mg per mL, in 0.6 N hydrochloric acid.
Water, Method Ib  921
921 :
between 4.5% and 5.5%, for hydrated Ephedrine; not more than 0.5% for anhydrous Ephedrine.
:
between 4.5% and 5.5%, for hydrated Ephedrine; not more than 0.5% for anhydrous Ephedrine.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chloride  221
221 —
A solution of 500 mg shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.030%).
—
A solution of 500 mg shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.030%).
Sulfate—
Dissolve 100 mg in 40 mL of water, and add 1 mL of 3 N hydrochloric acid and 1 mL of barium chloride TS: no turbidity develops within 10 minutes.
Ordinary impurities  466
466 —
—
Test solution:
methanol.
Standard solution:
methanol.
Eluant:
a mixture of isopropyl alcohol, ammonium hydroxide, and chloroform (80:15:5).
Visualization:
1, followed by 4.
Assay—
Dissolve about 500 mg of Ephedrine, accurately weighed, in 10 mL of neutralized alcohol, and add 5 drops of methyl red TS and 40.0 mL of 0.1 N hydrochloric acid VS. Titrate the excess acid with 0.1 N sodium hydroxide VS. Perform a blank determination (see Residual Titrations under Titrimetry  541
541 ). Each mL of 0.1 N hydrochloric acid is equivalent to 16.52 mg of C10H15NO.
). Each mL of 0.1 N hydrochloric acid is equivalent to 16.52 mg of C10H15NO.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 3049
Pharmacopeial Forum: Volume No. 30(3) Page 839