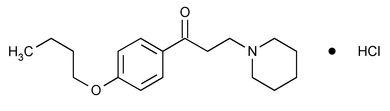
Dyclonine Hydrochloride
(dye' kloe neen hye'' droe klor' ide).
1-Propanone, 1-(4-butoxyphenyl)-3-(1-piperidinyl)-hydrochloride.
4¢-Butoxy-3-piperidinopropiophenone hydrochloride
» Dyclonine Hydrochloride contains not less than 98.0 percent and not more than 102.0 percent of C18H27NO2·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
B:
The retention time of the major peak in the chromatogram of the Assay preparation corresponds to that in the chromatogram of the Standard preparation, as obtained in the Assay.
C:
Add 2 mL of silver nitrate TS to 10 mL of Dyclonine Hydrochloride solution (1 in 100): a white precipitate is formed. Add 2 mL of nitric acid, centrifuge, and discard the supernatant. Wash the precipitate twice by adding 10 mL of 2 N nitric acid, centrifuging, and discarding the supernatant: the precipitate so obtained is soluble in 6 N ammonium hydroxide.
Melting range  741
741 :
between 173
:
between 173 and 178
and 178 .
.
pH  791
791 :
between 4.0 and 7.0, in a solution (1 in 100).
:
between 4.0 and 7.0, in a solution (1 in 100).
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 1 hour: it loses not more than 1.0% of its weight.
for 1 hour: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Assay—
Mobile phase, Standard preparation, and Chromatographic system—
Proceed as directed in the Assay under Dyclonine Hydrochloride Gel.
Assay preparation—
Transfer about 50 mg of Dyclonine Hydrochloride, accurately weighed, to a 500-mL volumetric flask, dissolve in 0.001 N phosphoric acid, dilute with 0.001 N phosphoric acid to volume, and mix.
Procedure—
Separately inject equal volumes (about 20 µL) of the Standard preparation and the Assay preparation into the chromatograph, record the chromatograms, and measure the responses for the major peaks. Calculate the quantity, in mg, of C18H27NO2·HCl in the portion of Dyclonine Hydrochloride taken by the formula:
500C(rU / rS)
in which C is the concentration, in mg per mL, of USP Dyclonine Hydrochloride RS in the Standard preparation; and rU and rS are the peak responses obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Domenick Vicchio, Ph.D.
Senior Scientific Liaison 1-301-998-6828 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2997
Pharmacopeial Forum: Volume No. 33(6) Page 1167