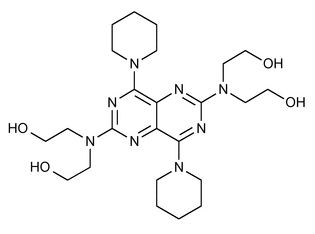
Dipyridamole
(dye'' pir id' a mole).
Ethanol, 2,2¢,2¢¢,2¢¢¢-[4,8-di-1-piperidinylpyrimido[5,4-d]pyrimidine-2,6-diyl)dinitrilo]tetrakis-.
2,2¢,2¢¢,2¢¢¢-[4,8-Dipiperidinopyrimido[5,4-d]pyrimidine-2,6-diyl)dinitrilo]tetraethanol
» Dipyridamole contains not less than 98.0 percent and not more than 102.0 percent of C24H40N8O4, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers. Store at room temperature.
Melting range  741
741 :
between 162
:
between 162 and 168
and 168 , but the range between beginning and end of melting does not exceed 2
, but the range between beginning and end of melting does not exceed 2 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 0.2% of its weight.
for 3 hours: it loses not more than 0.2% of its weight.
Chloride—
Dissolve 500 mg in 5 mL of alcohol and 2 mL of 2 N nitric acid, and add 1 mL of silver nitrate TS: no turbidity or precipitate is produced.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Chromatographic purity—
Mobile phase and Chromatographic system—
Prepare as directed in the Assay under Dipyridamole Tablets.
Test preparation A—
Prepare a solution of Dipyridamole in methanol having a known concentration of 1 mg per mL.
Test preparation B—
Dilute 1.0 mL of Test preparation A with methanol to 100 mL, and mix.
Procedure—
Inject 10 µL of Test preparation B into the chromatograph by means of a sampling valve, adjusting the operating parameters so that the response of the main peak (retention time about 6.5 minutes) obtained is about 5% full scale. Inject 10 µL of Test preparation A, and run the chromatograph for 10 minutes: the sum of responses of all secondary peaks obtained from Test preparation A is not greater than the response of the main peak obtained from Test preparation B (1.0%).
Assay—
Transfer about 450 mg of Dipyridamole, accurately weighed, to a 250-mL beaker, and dissolve in 50 mL of glacial acetic acid. Stir for 30 minutes. Add 75 mL of acetone, and stir for an additional 15 minutes. Titrate with 0.1 N perchloric acid VS, determining the endpoint potentiometrically, using a glass electrode and a silver-silver chloride reference electrode system. Perform a blank titration, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 50.46 mg of C24H40N8O4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Sujatha Ramakrishna, Ph.D.
Senior Scientific Liaison 1-301-816-8349 |
(SM22010) Monographs - Small Molecules 2 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2938
Pharmacopeial Forum: Volume No. 29(5) Page 1467