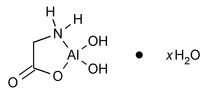
Dihydroxyaluminum Aminoacetate
(dye'' hye drox'' ee a loo' mi num a mee'' noe as' e tate).
Aluminum, (glycinato-N,O)dihydroxy-, hydrate.
(Glycinato)dihydroxyaluminum hydrate
Anhydrous 135.06
» Dihydroxyaluminum Aminoacetate yields not less than 94.0 percent and not more than 102.0 percent of dihydroxyaluminum aminoacetate (C2H6AlNO4), calculated on the dried basis. It may contain small amounts of aluminum oxide and of Aminoacetic Acid.
Packaging and storage—
Preserve in well-closed containers.
Identification—
Suspend 1 g in 25 mL of water, add hydrochloric acid, dropwise, until a clear solution is formed, and divide it into two equal parts for the following tests.
A:
One portion of the solution responds to the tests for Aluminum  191
191 .
.
B:
To the other portion of the solution add 1 drop of liquefied phenol and 5 mL of sodium hypochlorite TS: a blue color is produced.
pH  791
791 :
between 6.5 and 7.5, in a suspension of 1 g of it, finely powdered, in 25 mL of water.
:
between 6.5 and 7.5, in a suspension of 1 g of it, finely powdered, in 25 mL of water.
Loss on drying  731
731 —
Dry it at 130
—
Dry it at 130 to constant weight: it loses not more than 14.5% of its weight.
to constant weight: it loses not more than 14.5% of its weight.
Mercury, Method IIa  261
261 —
Transfer 2.0 g to a 100-mL beaker, and add 35 mL of 1 N sulfuric acid: the limit is 1 ppm.
—
Transfer 2.0 g to a 100-mL beaker, and add 35 mL of 1 N sulfuric acid: the limit is 1 ppm.
Isopropyl alcohol—
Transfer about 5 g to a flask provided with a reflux condenser, and add 100 mL of potassium permanganate solution (1 in 300) and 10 mL of sulfuric acid. Reflux the mixture for 30 minutes, distill, and collect 10 mL of the distillate. To 1 mL of the distillate add 5 drops of sodium nitroferricyanide TS and 2 mL of 1 N sodium hydroxide, then add a slight excess of 6 N acetic acid: no red color is produced.
Nitrogen—
Determine the nitrogen content as directed under Nitrogen Determination, Method II  461
461 , using about 100 mg, previously dried and accurately weighed. Each mL of 0.1 N sulfuric acid is equivalent to 1.401 mg of nitrogen. Not less than 9.90% and not more than 10.60% of nitrogen is found.
, using about 100 mg, previously dried and accurately weighed. Each mL of 0.1 N sulfuric acid is equivalent to 1.401 mg of nitrogen. Not less than 9.90% and not more than 10.60% of nitrogen is found.
Assay—
Edetate disodium titrant—
Prepare and standardize as directed in the Assay under Ammonium Alum.
Procedure—
Transfer about 2.5 g of Dihydroxyaluminum Aminoacetate, accurately weighed, to a 150-mL beaker, add 15 mL of hydrochloric acid, and warm, if necessary, to dissolve the specimen completely. Transfer the solution with the aid of water to a 500-mL volumetric flask, dilute with water to volume, and mix. Transfer 20.0 mL of this solution to a 250-mL beaker, and add, with continuous stirring, 25.0 mL of Edetate disodium titrant and then 20 mL of acetic acid–ammonium acetate buffer TS. Heat the solution near the boiling point for 5 minutes, cool, and add 50 mL of alcohol and 2 mL of dithizone TS. Titrate with 0.05 M zinc sulfate VS until the color changes from green-violet to rose pink. Perform a blank determination, substituting 20 mL of water for the assay solution, and make any necessary correction. Each mL of 0.05 M Edetate disodium titrant is equivalent to 6.753 mg of C2H6AlNO4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Elena Gonikberg, Ph.D.
Principal Scientific Liaison 1-301-816-8251 |
(SM32010) Monographs - Small Molecules 3 |
USP35–NF30 Page 2913