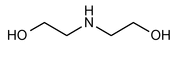
Diethanolamine
(dye'' eth a nol' a meen).
» Diethanolamine is a mixture of ethanolamines, consisting largely of diethanolamine. It contains not less than 98.5 percent and not more than 101.0 percent of ethanolamines, calculated on the anhydrous basis as NH(C2H4OH)2.
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification,
Infrared Absorption  197F
197F .
.
Refractive index  831
831 :
between 1.473 and 1.476, at 30
:
between 1.473 and 1.476, at 30 .
.
Water, Method I  921
921 :
not more than 0.15%, a 20-g test specimen being used, and a mixture of 25 mL of glacial acetic acid and 40 mL of methanol being used as the solvent.
:
not more than 0.15%, a 20-g test specimen being used, and a mixture of 25 mL of glacial acetic acid and 40 mL of methanol being used as the solvent.
Limit of triethanolamine—
Mixed indicator—
Dissolve 0.15 g of methyl orange and 0.08 g of xylene cyanole FF in 100 mL of water, and mix.
Procedure—
Place 100 mL of methanol and 6 to 8 drops of Mixed indicator in a 500-mL glass-stoppered conical flask, and neutralize with 0.1 N alcoholic sulfuric acid or 0.1 N alcoholic potassium hydroxide. The neutral solution is amber when viewed by transmitted light and is red-brown when viewed by reflected light. Add about 20 g of test specimen, accurately weighed. Cautiously add 75 mL of acetic anhydride, and swirl to effect complete solution. Allow to stand at room temperature for 30 minutes. Cool to room temperature, if necessary. Titrate with 0.5 N alcoholic sulfuric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.5 N alcoholic sulfuric acid is equivalent to 74.6 mg of triethanolamine: the limit is 1.0%, by weight.
Assay—
Transfer about 2 g of Diethanolamine, accurately weighed, to a 250-mL conical flask. Add 50 mL of water and bromocresol green TS, and titrate with 0.5 N hydrochloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.5 N hydrochloric acid is equivalent to 52.57 mg of diethanolamine, expressed as NH(C2H4OH)2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Robert H. Lafaver, M.S.
Scientific Liaison 1-301-816-8335 |
(EXC2010) Monographs - Excipients |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 1783
Pharmacopeial Forum: Volume No. 31(5) Page 1422