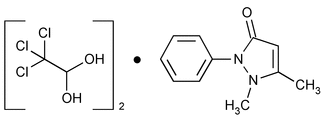
Dichloralphenazone
(dye'' klor al fen' a zone).
1,2-Dihydro-1,5-dimethyl-2-phenyl-3H-pyrazol-3-one, compound with 2,2,2-trichloro-1,1-ethanediol (1:2).
Antipyrine, compound with chloral hydrate (1:2)
» Dichloralphenazone contains not less than 97.0 percent and not more than 100.5 percent of C15H18Cl6N2O5, determined by both Assay procedures.
Packaging and storage—
Preserve in well-closed containers.
USP Reference standards  11
11 —
—
USP Dichloralphenazone RS
Identification—
B:
Dissolve 0.1 g of it in 10 mL of water containing 0.1 g of sodium nitrite, and add 1 mL of 2 N sulfuric acid: a green color is produced.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Assay—
Perform the following determinations.
Assay procedure 1—
Transfer about 1500 mg of Dichloralphenazone, accurately weighed, to a beaker, add 10 mL of water and 20.0 mL of 0.5 N sodium hydroxide VS. Allow to stand for 2 minutes, and titrate with 0.5 N sulfuric acid VS to a colorless phenolphthalein endpoint. Titrate this neutralized solution with 0.1 N silver nitrate VS, using potassium chromate TS as the indicator. To the volume of 0.5 N sulfuric acid used in the first titration add four-fifteenths of the volume of 0.1 N silver nitrate used in the second titration, and subtract the sum obtained from the volume of 0.5 N sodium hydroxide added. Each mL of 0.5 N sodium hydroxide represented by the difference is equivalent to 129.8 mg of C15H18Cl6N2O5.
Assay procedure 2—
Transfer about 400 mg of Dichloralphenazone, accurately weighed, to a 125-mL conical flask, add 20 mL of a 1 in 10 solution of sodium acetate, and swirl to dissolve. Add 25.0 mL of 0.1 N iodine VS, and allow to stand in the dark for 20 minutes, gently swirling occasionally. Add 10 mL of chloroform, rinsing the walls of the flask, and swirl until the precipitate is dissolved. Titrate the excess iodine with 0.1 N sodium thiosulfate VS, adding 3 mL of starch TS as the endpoint is approached. Perform a blank titration (see Residual Titrations under Titrimetry  541
541 ). The difference between the titrations represents the amount of iodine consumed. Each mL of 0.1 N iodine consumed is equivalent to 25.95 mg of C15H18Cl6N2O5.
). The difference between the titrations represents the amount of iodine consumed. Each mL of 0.1 N iodine consumed is equivalent to 25.95 mg of C15H18Cl6N2O5.
Calculation—
The difference between the percentages calculated from Assay procedure 1 and Assay procedure 2 is not more than 1.5%.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2876