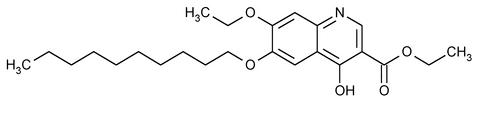
Decoquinate
(dee'' koe kwin' ate).
3-Quinolinecarboxylic acid, 6-(decyloxy)-7-ethoxy-4-hydroxy-, ethyl ester.
Ethyl 6-(decyloxy)-7-ethoxy-4-hydroxy-3-quinolinecarboxylate
» Decoquinate contains not less than 99.0 percent and not more than 101.0 percent of C24H35NO5, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Labeling—
Label it to indicate that it is for veterinary use only.
Identification—
B:
Transfer about 40 mg of it, accurately weighed, to a 100-mL volumetric flask, add 10 mL of hot chloroform, swirl to dissolve, and while still warm add about 60 mL of dehydrated alcohol. Allow to cool, dilute with dehydrated alcohol to volume, and mix. Promptly transfer 10.0 mL of this solution to a second 100-mL volumetric flask, dilute with dehydrated alcohol to volume, and mix. Transfer 10.0 mL of this solution to a third 100-mL volumetric flask, add 10 mL of 0.1 N hydrochloric acid, dilute with dehydrated alcohol to volume, and mix: the UV absorption spectrum of this solution exhibits maxima and minima at the same wavelengths as that of a similar solution of USP Decoquinate RS, concurrently measured, and the respective absorptivities, calculated on the dried basis, at the wavelength of maximum absorption at about 265 nm do not differ by more than 2.5%.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 to constant weight: it loses not more than 0.5% of its weight.
to constant weight: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Ordinary impurities  466
466 —
—
Test solution:
chloroform, prepared with the aid of heat.
Standard solution:
chloroform, using dilutions of the Test solution.
Eluant:
a mixture of chloroform, dehydrated alcohol, and anhydrous formic acid (85:10:5).
Visualization:
1.
Tolerances:
no impurity exceeds 1%, and the total does not exceed 2%.
Assay—
Dissolve about 1000 mg of Decoquinate, accurately weighed, in 100 mL of glacial acetic acid, with the aid of gentle heat. Allow to cool, add 1 drop of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 41.76 mg of C24H35NO5.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Morgan Puderbaugh, B.S.
Associate Scientific Liaison 1-301-998-6833 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2812