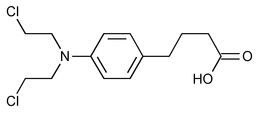Chlorambucil
(klor am' bue sil).
Benzenebutanoic acid, 4-[bis(2-chloroethyl)amino]-.
4-[p-[Bis(2-chloroethyl)amino]phenyl]butyric acid
» Chlorambucil contains not less than 98.0 percent and not more than 101.0 percent of C14H19Cl2NO2, calculated on the anhydrous basis.
[Caution—Great care should be taken to prevent inhaling particles of Chlorambucil and exposing the skin to it.
]
Packaging and storage—
Preserve in tight, light-resistant containers.
Identification—
A:
Infrared Absorption  197S
197S : 1 in 125 solution, carbon disulfide, 1-mm cell.
: 1 in 125 solution, carbon disulfide, 1-mm cell.
B:
Dissolve 50 mg in 5 mL of acetone, and dilute with water to 10 mL. Add 1 drop of 2 N sulfuric acid, then add 4 drops of silver nitrate TS: no opalescence is observed immediately (absence of chloride ion). Warm the solution on a steam bath: opalescence develops (presence of ionizable chlorine).
Melting range  741
741 :
between 65
:
between 65 and 69
and 69 .
.
Water, Method I  921
921 :
not more than 0.5%.
:
not more than 0.5%.
Assay—
Dissolve about 200 mg of Chlorambucil, accurately weighed, in 10 mL of acetone, add 10 mL of water, and titrate with 0.1 N sodium hydroxide VS, using phenolphthalein TS as the indicator. Each mL of 0.1 N sodium hydroxide is equivalent to 30.42 mg of C14H19Cl2NO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Feiwen Mao, M.S.
Senior Scientific Liaison 1-301-816-8320 |
(SM32010) Monographs - Small Molecules 3 |
| Reference Standards | RS Technical Services 1-301-816-8129 rstech@usp.org |
USP35–NF30 Page 2605