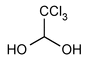Chloral Hydrate
(klor' al hye' drate).
» Chloral Hydrate contains not less than 99.5 percent and not more than 102.5 percent of C2H3Cl3O2.
Packaging and storage—
Preserve in tight containers.
Identification—
Transfer to a 125-mL conical flask a portion of a solution in water equivalent to about 1 mg of chloral hydrate, and add water to bring the volume to about 10 mL. Add 10 mL of 1-ethylquinaldinium iodide solution (15 in 1000), which has been filtered through a 0.45-µm filter. Add 60 mL of isopropyl alcohol, 5 mL of an aqueous 0.1 M monoethanolamine solution, and 15 mL of water. Mix, and heat in a water bath at 60 for 15 minutes: a blue color develops.
for 15 minutes: a blue color develops.
Acidity—
A 1 in 20 solution in alcohol does not at once redden moistened blue litmus paper.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Chloride  221
221 —
To a 1 in 10 solution in alcohol add a few drops of silver nitrate TS: any opalescence produced does not exceed that of a control containing 0.10 mL of 0.020 N hydrochloric acid (0.007%).
—
To a 1 in 10 solution in alcohol add a few drops of silver nitrate TS: any opalescence produced does not exceed that of a control containing 0.10 mL of 0.020 N hydrochloric acid (0.007%).
Readily carbonizable substances  271
271 —
Shake 500 mg, at intervals of 5 minutes during 1 hour, with 5 mL of sulfuric acid in a glass-stoppered cylinder that previously has been rinsed with sulfuric acid, and transfer the mixture to a comparison vessel: the mixture has no more color than Matching Fluid P.
—
Shake 500 mg, at intervals of 5 minutes during 1 hour, with 5 mL of sulfuric acid in a glass-stoppered cylinder that previously has been rinsed with sulfuric acid, and transfer the mixture to a comparison vessel: the mixture has no more color than Matching Fluid P.
Assay—
Dissolve about 4 g of Chloral Hydrate, accurately weighed, in 10 mL of water, add 30.0 mL of 1 N sodium hydroxide VS, and allow the mixture to stand for 2 minutes. Add a few drops of phenolphthalein TS, and titrate the residual alkali at once with 1 N sulfuric acid VS. Each mL of 1 N sodium hydroxide corresponds to 165.4 mg of C2H3Cl3O2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
|---|---|---|
| Monograph | Ravi Ravichandran, Ph.D.
Principal Scientific Liaison 1-301-816-8330 |
(SM42010) Monographs - Small Molecules 4 |
USP35–NF30 Page 2604
Pharmacopeial Forum: Volume No. 34(5) Page 1150