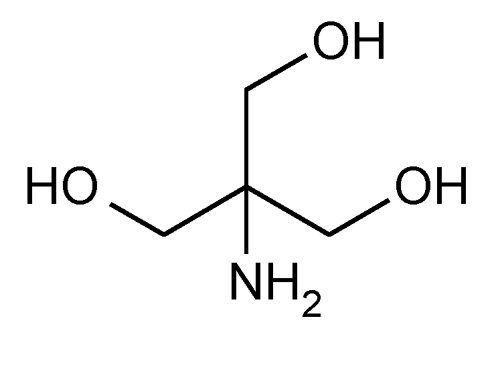
Tromethamine
1,3-Propanediol, 2-amino-2-(hydroxymethyl)-.
2-Amino-2-(hydroxymethyl)-1,3-propanediol
» Tromethamine contains not less than 99.0 percent and not more than 101.0 percent of C4H11NO3, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
B:
To 4.5 mL of a saturated solution of salicylaldehyde add 0.5 mL of glacial acetic acid, and mix. Add 4.0 mL of a solution of Tromethamine (1 in 5), and mix: a yellow color is produced.
C:
To 0.5 mL of a 4 in 10 solution of ceric ammonium nitrate in 2 N nitric acid add 3 mL of water and 0.5 mL of a solution of Tromethamine (1 in 5), and mix: the color changes from light yellow to orange.
pH  791
791 :
between 10.0 and 11.5, in a solution (1 in 20).
:
between 10.0 and 11.5, in a solution (1 in 20).
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 3 hours: it loses not more than 1.0% of its weight.
for 3 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Assay—
Dissolve about 250 mg of Tromethamine, accurately weighed, in 100 mL of water, add bromocresol purple TS, and titrate with 0.1 N hydrochloric acid VS to a yellow endpoint. Each mL of 0.1 N hydrochloric acid is equivalent to 12.11 mg of C4H11NO3.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Feiwen Mao, M.S.
Scientist 1-301-816-8320 |
(MDOOD05) Monograph Development-Ophthalmics Oncologics and Dermatologicals |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3822
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.