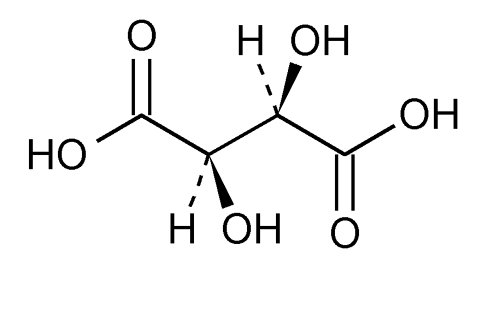
Tartaric Acid
Butanedioic acid, 2,3-dihydroxy-; Butanedioic acid, 2,3-dihydroxy-, [R-(R*,R*)]-.
Tartaric acid; l -(+)-Tartaric acid
» Tartaric Acid, dried over phosphorus pentoxide for 3 hours, contains not less than 99.7 percent and not more than 100.5 percent of C4H6O6.
Packaging and storage—
Preserve in well-closed containers.
Identification—
A:
It responds to the tests for Tartrate  191
191 .
.
B:
When ignited, it gradually decomposes, emitting an odor resembling that of burning sugar (distinction from citric acid).
Loss on drying  731
731 —
Dry it over phosphorus pentoxide for 3 hours: it loses not more than 0.5% of its weight.
—
Dry it over phosphorus pentoxide for 3 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Limit of oxalate—
Nearly neutralize 10 mL of a solution of it (1 in 10) with 6 N ammonium hydroxide, and add 10 mL of calcium sulfate TS: no turbidity is produced.
Sulfate  221
221 —
To 10 mL of a solution (1 in 100) add 3 drops of hydrochloric acid and 1 mL of barium chloride TS: no turbidity is produced.
—
To 10 mL of a solution (1 in 100) add 3 drops of hydrochloric acid and 1 mL of barium chloride TS: no turbidity is produced.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Assay—
Place about 2 g of Tartaric Acid, previously dried and accurately weighed, in a conical flask. Dissolve it in 40 mL of water, add phenolphthalein TS, and titrate with 1 N sodium hydroxide VS. Each mL of 1 N sodium hydroxide is equivalent to 75.04 mg of C4H6O6.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Robert H. Lafaver, B.A.
Scientist 1-301-816-8335 |
(EM105) Excipient Monographs 1 |
USP32–NF27 Page 1368
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.