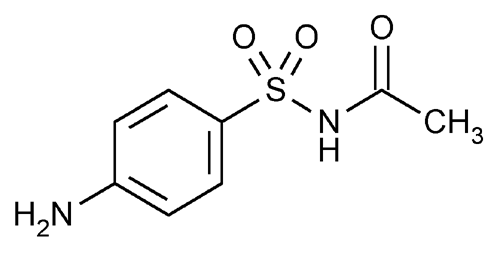
Sulfacetamide
» Sulfacetamide contains not less than 99.0 percent and not more than 100.5 percent of C8H10N2O3S, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed, light-resistant containers.
Clarity and color of solution—
Dissolve about 200 mg in 5 mL of 1 N sodium hydroxide: a yellow to faintly yellow solution having not more than a trace of turbidity is produced.
Identification—
B:
Place about 500 mg in a test tube, heat gently until it boils, and cool: an oily liquid, which has the characteristic odor of acetamide, condenses on the walls of the test tube (distinction from the sublimates of sulfadiazine, sulfamerazine, sulfamethazine, and sulfapyrazine, which are solids at room temperature).
Reaction—
A solution (1 in 150) is acid to litmus.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 2 hours: it loses not more than 0.5% of its weight.
for 2 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Sulfate  221
221 —
Digest 1 g with 50 mL of water at about 70
—
Digest 1 g with 50 mL of water at about 70 for 5 minutes. Cool immediately to room temperature, and filter. A 25-mL portion of the filtrate so obtained shows no more sulfate than corresponds to 0.2 mL of 0.02 N sulfuric acid (0.04%).
for 5 minutes. Cool immediately to room temperature, and filter. A 25-mL portion of the filtrate so obtained shows no more sulfate than corresponds to 0.2 mL of 0.02 N sulfuric acid (0.04%).
Selenium  291
291 :
0.003%, a 200-mg test specimen being used.
:
0.003%, a 200-mg test specimen being used.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Assay—
Proceed with Sulfacetamide as directed under Nitrite Titration  451
451 . Each mL of 0.1 M sodium nitrite is equivalent to 21.42 mg of C8H10N2O3S.
. Each mL of 0.1 M sodium nitrite is equivalent to 21.42 mg of C8H10N2O3S.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientist 1-301-816-8394 |
(MDAA05) Monograph Development-Antivirals and Antimicrobials |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3613