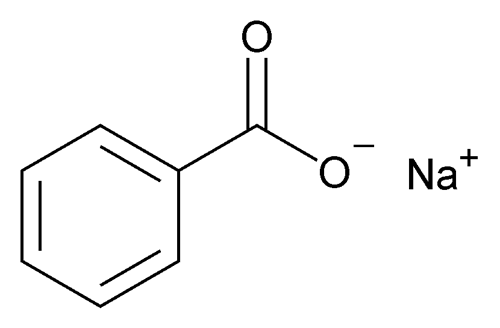
Sodium Benzoate
» Sodium Benzoate contains not less than 99.0 percent and not more than 100.5 percent of C7H5NaO2, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
A:
Infrared Absorption  197M
197M , on the undried specimen.
, on the undried specimen.
Alkalinity—
Dissolve 2 g in 20 mL of hot water, and add 2 drops of phenolphthalein TS: the pink color produced, if any, is discharged by the addition of 0.20 mL of 0.10 N sulfuric acid.
Water, Method I  921
921 :
not more than 1.5%.
:
not more than 1.5%.
Heavy metals  231
231 —
Dissolve 4.0 g in 40 mL of water, add, dropwise, with vigorous stirring, 10 mL of 3 N hydrochloric acid, and filter. Use 25 mL of the filtrate: the limit is 0.001%.
—
Dissolve 4.0 g in 40 mL of water, add, dropwise, with vigorous stirring, 10 mL of 3 N hydrochloric acid, and filter. Use 25 mL of the filtrate: the limit is 0.001%.
Assay—
Transfer about 600 mg of Sodium Benzoate, accurately weighed, to a 250-mL beaker. Add 100 mL of glacial acetic acid, stir until the assay specimen is completely dissolved, add 2 drops of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 14.41 mg of C7H5NaO2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Robert H. Lafaver, B.A.
Scientist 1-301-816-8335 |
(EM105) Excipient Monographs 1 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1339
Pharmacopeial Forum: Volume No. 31(3) Page 818
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.