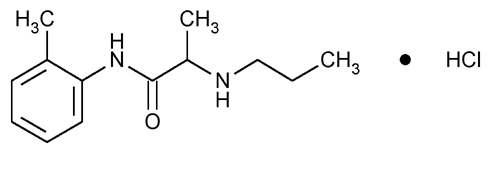
Prilocaine Hydrochloride
Propanamide, N-(2-methylphenyl)-2-(propylamino)-, monohydrochloride.
2-(Propylamino)-o-propionotoluidide monohydrochloride
» Prilocaine Hydrochloride contains not less than 99.0 percent and not more than 101.0 percent of C13H20N2O·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
B:
Dissolve about 300 mg in 5 mL of water, add 4 mL of 6 N ammonium hydroxide, and extract with 50 mL of chloroform. Filter the extract, and evaporate the filtrate on a steam bath with the aid of a current of air. Dissolve about 100 mg of the prilocaine so obtained in 1 mL of alcohol, add 10 drops of cobaltous chloride TS, and shake for 2 minutes: a bright green color develops, and a precipitate is formed.
C:
Dissolve about 100 mg in 3 mL of water, render the solution alkaline with 6 N ammonium hydroxide, and filter: the filtrate responds to the tests for Chloride  191
191 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 0.3% of its weight.
for 4 hours: it loses not more than 0.3% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Heavy metals, Method I  231
231 :
0.002%.
:
0.002%.
Assay—
Dissolve about 500 mg of Prilocaine Hydrochloride, accurately weighed, in 50 mL of glacial acetic acid, add 10 mL of mercuric acetate TS and 2 drops of crystal violet TS, and titrate with 0.1 N perchloric acid VS to a blue-green endpoint. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 25.68 mg of C13H20N2O·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3379
Pharmacopeial Forum: Volume No. 29(5) Page 1564