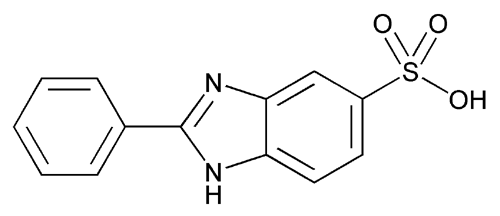
Ensulizole
1H-Benzimidazole-5-sulfonic acid-2-phenyl-.
2-Phenylbenzimidazole-5-sulfonic acid
» Ensulizole contains not less than 98.0 percent and not more than 102.0 percent of C13H10N2O3S, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers in a cool place.
Identification—
Solution:
5 µg per mL, prepared as follows. Transfer about 100 mg of Ensulizole, accurately weighed, to a 100-mL volumetric flask, dissolve in 4 mL of 1 N sodium hydroxide, and dilute with water to volume. Transfer 0.5 mL of the solution to a 100-mL volumetric flask, dilute with water to volume, and mix.
Absorptivities, calculated on the dried basis, at the wavelength of maximum absorbance at about 302 nm, do not differ by more than 3.0%.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 2.0% of its weight.
for 4 hours: it loses not more than 2.0% of its weight.
Assay—
Transfer about 1200 mg of Ensulizole, accurately weighed, to a 300-mL iodine flask, and dissolve by stirring in 25 mL of 0.5 N sodium hydroxide. Add phenolphthalein TS, and titrate the excess with 0.5 N hydrochloric acid VS. Perform a blank determination, and make any necessary correction. Each mL of 0.5 N sodium hydroxide is equivalent to 137.15 mg of C13H10N2O3S. Calculate the percentage of C13H10N2O3S in the portion of Ensulizole taken by the formula:
in which NHCl is the actual normality of 0.5 N hydrochloric acid; VB is the volume, in mL, of 0.5 N hydrochloric acid used in the titration of the blank; VA is the volume, in mL, of 0.5 N hydrochloric acid used in the titration of the sample; W is the weight, in mg, of Ensulizole taken; and LOD is the percentage of loss on drying expressed as a decimal fraction, as determined in the test for Loss on drying.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientist 1-301-816-8394 |
(MDAA05) Monograph Development-Antivirals and Antimicrobials |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2257
Pharmacopeial Forum: Volume No. 32(6) Page 1677