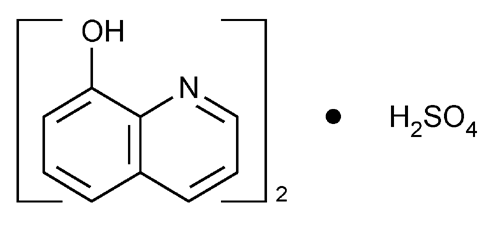
Oxyquinoline Sulfate
8-Quinolinol sulfate (2:1) (salt).
8-Quinolinol sulfate (2:1) (salt)
» Oxyquinoline Sulfate is 8-hydroxyquinoline sulfate. It contains not less than 97.0 percent and not more than 101.0 percent of (C9H7NO)2·H2SO4, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
A:
Infrared Absorption  197M
197M , on undried specimen.
, on undried specimen.
B:
A solution (1 in 10) responds to the tests for Sulfate  191
191 .
.
Water, Method I  921
921 :
between 4.0% and 6.0%.
:
between 4.0% and 6.0%.
Residue on ignition  281
281 :
not more than 0.3%.
:
not more than 0.3%.
Heavy metals, Method II  231
231 :
20 µg per g.
:
20 µg per g.
Assay—
Transfer about 100 mg of Oxyquinoline Sulfate, accurately weighed, to an iodine flask, add 30 mL of glacial acetic acid, 25.0 mL of 0.1 N bromine VS, 10 mL of potassium bromide solution (3 in 20), and 10 mL of hydrochloric acid, immediately insert the stopper, mix, and allow to stand for 15 minutes, protected from light. Quickly add 10 mL of potassium iodide solution (1 in 10) and 100 mL of water, taking precautions against the escape of bromine vapor, at once insert the stopper, and shake the mixture thoroughly. Remove the stopper, and rinse it and the neck of the flask with a small quantity of water so that the washing flows into the flask. Shake the mixture thoroughly, and titrate the liberated iodine with 0.1 N sodium thiosulfate VS, adding 3 mL of starch TS as the endpoint is approached. Perform a blank determination (see Residual Titrations under Titrimetry  541
541 ). Each mL of 0.1 N bromine is equivalent to 4.855 mg of (C9H7NO)2·H2SO4.
). Each mL of 0.1 N bromine is equivalent to 4.855 mg of (C9H7NO)2·H2SO4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Robert H. Lafaver, B.A.
Scientist 1-301-816-8335 |
(EM105) Excipient Monographs 1 |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1294
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.