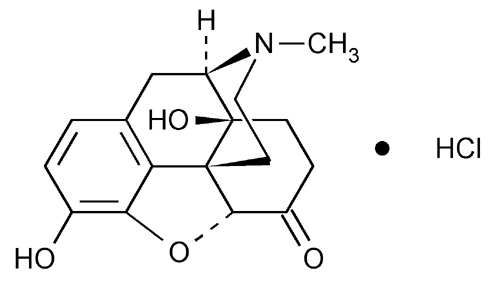
Oxymorphone Hydrochloride
Morphinan-6-one, 4,5-epoxy-3,14-dihydroxy-17-methyl-, hydrochloride, (5
4,5
» Oxymorphone Hydrochloride contains not less than 97.0 percent and not more than 102.0 percent of C17H19NO4·HCl, calculated on the dried basis.
Packaging and storage—
Preserve in tight, light-resistant containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification—
A:
Dissolve about 250 mg in 25 mL of water, and render the solution alkaline with a saturated solution of sodium bicarbonate. Extract the liberated oxymorphone with two 15-mL portions of chloroform. Reserve the chloroform extracts, combined in a second separator, for Identification test B: the aqueous phase, acidified with 2 N nitric acid, responds to the tests for Chloride  191
191 .
.
B:
Wash the combined chloroform extracts from Identification test A with 5 mL of water, and filter. Evaporate the chloroform solution on a steam bath nearly to dryness, then add a few mL of ether, and continue the evaporation with stirring until the solvent is removed: the IR absorption spectrum of a 1 in 50 solution in alcohol-free chloroform of the oxymorphone so obtained, determined in a 0.5-mm cell, exhibits maxima only at the same wavelengths as that of a similar solution of USP Oxymorphone RS.
C:
The UV absorption spectrum of a 1 in 6500 solution in 0.1 N hydrochloric acid exhibits maxima and minima at the same wavelengths as that of a solution of USP Oxymorphone RS, prepared by dissolving about 20 mg of the Reference Standard in 10 mL of 1 N hydrochloric acid and diluting with water to 100.0 mL. The ratio A281 / A264 is 1.75 ± 0.2.
D:
Dissolve 10 mg in 1 mL of water, and add a few drops of ferric chloride TS: a blue color is produced immediately.
Acidity—
Dissolve 300 mg in 10 mL of water, add 1 drop of methyl red TS, and titrate with 0.020 N sodium hydroxide: not more than 0.30 mL is required to produce a yellow color.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 18 hours: it loses not more than 8.0% of its weight.
for 18 hours: it loses not more than 8.0% of its weight.
Residue on ignition  281
281 :
not more than 0.3%.
:
not more than 0.3%.
Limit of nonphenolic substances—
Dissolve 1 g in 15 mL of water, add 5 mL of sodium hydroxide solution (2 in 25), and extract with three 10-mL portions of chloroform. Filter the combined extracts through a small chloroform-moistened filter paper, and wash the filtrate with 5 mL of water. Filter the chloroform layer through chloroform-moistened filter paper into a tared, 50-mL beaker, and evaporate on a steam bath with the aid of a gentle current of filtered air to dryness. Dry the beaker and residue at 105 for 1 hour, and weigh: the residue so obtained does not exceed 15 mg.
for 1 hour, and weigh: the residue so obtained does not exceed 15 mg.
Ordinary impurities  466
466 —
—
Test solution:
methanol.
Standard solution:
methanol.
Eluant:
a mixture of dehydrated alcohol, cyclohexane, and ammonium hydroxide (10:5:1).
Visualization:
1.
Chloride content—
Dissolve about 300 mg, accurately weighed, in 50 mL of methanol in a glass-stoppered flask, add 5 mL of glacial acetic acid and 3 drops of eosin Y TS, and titrate with 0.1 N silver nitrate VS. Each mL of 0.1 N silver nitrate is equivalent to 3.545 mg of Cl: the content is between 10.2% and 10.8%, calculated on the dried basis.
Assay—
Transfer about 700 mg of Oxymorphone Hydrochloride, accurately weighed, to a glass-stoppered flask containing 50 mL of glacial acetic acid and 10 mL of mercuric acetate TS. Add 3 mL of acetic anhydride and 1 drop of methyl violet TS, and titrate with 0.1 N perchloric acid VS to a clear blue color. Perform a blank determination, and make any necessary correction. Each mL of 0.1 N perchloric acid is equivalent to 33.78 mg of C17H19NO4·HCl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Clydewyn M. Anthony, Ph.D.
Scientist 1-301-816-8139 |
(MDCCA05) Monograph Development-Cough Cold and Analgesics |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 3176
Pharmacopeial Forum: Volume No. 29(6) Page 1946