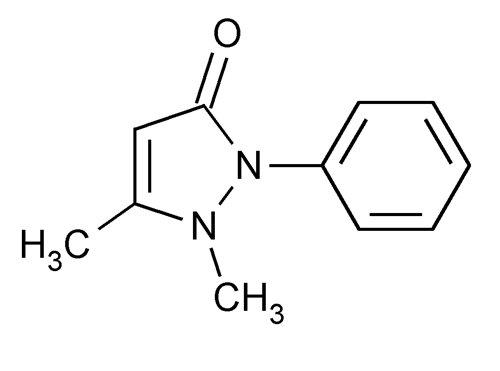
Antipyrine
1,2-Dihydro-1,5-dimethyl-2-phenyl-3H-pyrazol-3-one.
2,3-Dimethyl-1-phenyl-3-pyrazolin-5-one
» Antipyrine contains not less than 99.0 percent and not more than 100.5 percent of C11H12N2O, calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Completeness and color of solution—
It is completely soluble in its own weight of cold water, the solution being colorless or not more than slightly yellow when viewed transversely in a tube having a diameter of about 20 mm.
Identification—
B:
Ultraviolet Absorption  197U
197U —
—
Solution:
20 µg per mL.
Medium:
methanol.
Absorptivities at 266 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
Add tannic acid TS to a solution of it: a white precipitate is formed.
Loss on drying  731
731 —
Dry it at 60
—
Dry it at 60 for 2 hours: it loses not more than 1.0% of its weight.
for 2 hours: it loses not more than 1.0% of its weight.
Residue on ignition  281
281 :
not more than 0.15%.
:
not more than 0.15%.
Heavy metals  231
231 —
Dissolve 1 g in 2 mL of 1 N acetic acid, and add water to make 25 mL: the limit is 0.002%.
—
Dissolve 1 g in 2 mL of 1 N acetic acid, and add water to make 25 mL: the limit is 0.002%.
Ordinary impurities  466
466 —
—
Test solution:
chloroform.
Standard solution:
chloroform.
Eluant:
a mixture of chloroform, acetone, butyl alcohol, and formic acid (60:15:15:15).
Visualization:
1.
Assay—
Transfer about 150 mg of Antipyrine, accurately weighed, to a 250-mL iodine flask, and dissolve in 25 mL of water. Add 2 g of sodium acetate, 1 mL of diluted acetic acid, and 20.0 mL of 0.1 N iodine VS, mix, and allow to stand in a cool, dark place for 20 minutes. Add 25 mL of alcohol to dissolve the precipitate, and titrate the excess iodine with 0.1 N sodium thiosulfate VS, using starch TS as the indicator. Each mL of 0.1 N iodine is equivalent to 9.412 mg of C11H12N2O.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Clydewyn M. Anthony, Ph.D.
Scientist 1-301-816-8139 |
(MDCCA05) Monograph Development-Cough Cold and Analgesics |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1569