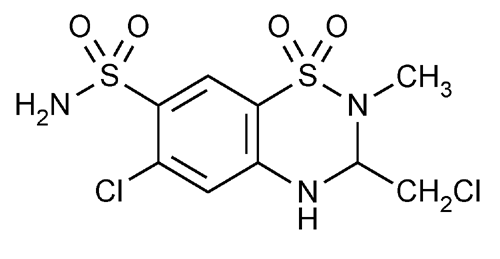
Methyclothiazide
2H-1,2,4-Benzothiadiazine-7-sulfonamide, 6-chloro-3-(chloromethyl)-3,4-dihydro-2-methyl-, 1,1-dioxide, (±)-.
(±)-6-Chloro-3-(chloromethyl)-3,4-dihydro-2-methyl-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide
» Methyclothiazide contains not less than 97.0 percent and not more than 102.0 percent of C9H11Cl2N3O4S2, calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
USP Reference standards  11
11 —
—
USP Methyclothiazide RS.
USP Methyclothiazide Related Compound A RS.
USP Methyclothiazide RS.
USP Methyclothiazide Related Compound A RS.
Identification—
Solution:
20 µg per mL.
Medium:
methanol.
C:
Fuse about 100 mg of it with a pellet of sodium hydroxide: the ammonia fumes produced turn moistened red litmus paper blue. The fused mixture responds to the test for Sulfite  191
191 .
.
Loss on drying  731
731 —
Dry it at 105
—
Dry it at 105 for 4 hours: it loses not more than 0.5% of its weight.
for 4 hours: it loses not more than 0.5% of its weight.
Residue on ignition  281
281 :
not more than 0.2%.
:
not more than 0.2%.
Chloride  221
221 —
Shake 750 mg with 25 mL of water for 2 minutes, filter through a suitable membrane filter, and add 4 or 5 drops of 2 N nitric acid: the acidified filtrate shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.02%).
—
Shake 750 mg with 25 mL of water for 2 minutes, filter through a suitable membrane filter, and add 4 or 5 drops of 2 N nitric acid: the acidified filtrate shows no more chloride than corresponds to 0.20 mL of 0.020 N hydrochloric acid (0.02%).
Selenium  291
291 :
0.003%.
:
0.003%.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Diazotizable substances—
Standard preparation—
Transfer about 10 mg of USP Methyclothiazide Related Compound A RS, accurately weighed, to a 50-mL volumetric flask, dilute with acetonitrile to volume, and mix. Pipet 25 mL of the solution into a 100-mL volumetric flask, dilute with acetonitrile to volume, and mix. Each mL of Standard preparation contains about 50 µg of the Reference Standard.
Test preparation—
Transfer about 500 mg of Methyclothiazide, accurately weighed, to a 100-mL volumetric flask, dissolve in and dilute with acetonitrile to volume, and mix.
Procedure—
Pipet 2 mL each of the Standard preparation and the Test preparation into separate 50-mL volumetric flasks. Pipet 2 mL of acetonitrile into a third 50-mL flask to provide the blank. To each flask add 4 mL of 0.1 N hydrochloric acid, and mix. Add 3.0 mL of sodium nitrite solution (1 in 200) to each flask, mix, and place the flasks in an ice bath for 5 minutes, shaking occasionally. Add to each flask 3.0 mL of ammonium sulfamate solution (1 in 50), mix, and allow the flasks to remain in the ice bath for 1 additional minute. Remove the flasks from the ice bath, add 1.0 mL of N-(1-naphthyl)ethylenediamine dihydrochloride solution (1 in 1000), and mix. Allow the flasks to stand at room temperature for 1 minute, then dilute with water to volume, and mix. Concomitantly determine the absorbances of the solutions obtained from the Standard preparation and the Test preparation in 1-cm cells at 525 nm, with a suitable spectrophotometer, using the reagent blank to set the instrument. The absorbance of the solution from the Test preparation does not exceed that of the solution from the Standard preparation, corresponding to not more than 1.0% of diazotizable substances.
Assay—
Transfer about 350 mg of Methyclothiazide, accurately weighed, to a 250-mL conical flask, add 40 mL of a 1 in 20 solution of potassium hydroxide in methanol, and reflux at full boil for 1 hour. Cool, rinse the inner walls of the condenser with 20 mL of water and two 20-mL portions of methanol, add 10 mL of glacial acetic acid and 2 drops of eosin Y TS, and titrate with 0.1 N silver nitrate VS to the first appearance of a definite pink color. Each mL of 0.1 N silver nitrate is equivalent to 36.02 mg of C9H11Cl2N3O4S2.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Sujatha Ramakrishna, Ph.D.
Scientist 1-301-816-8349 |
(MDCV05) Monograph Development-Cardiovascular |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2936