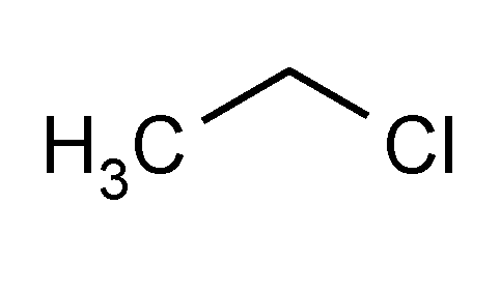
Ethyl Chloride
» Ethyl Chloride contains not less than 99.5 percent and not more than 100.5 percent of C2H5Cl.
Packaging and storage—
Preserve in tight containers, preferably hermetically sealed, and remote from fire.
Reaction—
Shake 10 mL with 10 mL of water, both having been previously cooled to 0 , and allow the supernatant layer of ethyl chloride to volatilize spontaneously: the remaining liquid is neutral to litmus. Retain the liquid for the test for Alcohol.
, and allow the supernatant layer of ethyl chloride to volatilize spontaneously: the remaining liquid is neutral to litmus. Retain the liquid for the test for Alcohol.
Limit of nonvolatile residue and odor—
Allow 5 mL to evaporate spontaneously from a tared, shallow dish: no foreign odor is perceptible while the last portions evaporate, and the weight of the residue is negligible.
Chloride—
Add a few drops of silver nitrate TS to 10 mL of alcohol, cool to 0 , and add to the clear liquid about 500 µL of Ethyl Chloride cooled to the same temperature: no turbidity is produced immediately.
, and add to the clear liquid about 500 µL of Ethyl Chloride cooled to the same temperature: no turbidity is produced immediately.
Assay—
Introduce about 1.5 mL of cold Ethyl Chloride into a tared glass-stoppered pressure bottle containing 25.0 mL of 1 N alcoholic potassium hydroxide VS, rapidly replace the stopper, and weigh accurately. Tie down the stopper, insert the bottle in a wire basket, and immerse in a water bath at room temperature. [Caution—Before raising the bath temperature, take adequate precautions to cover the bottle or erect a suitable safety shield to prevent injury in case the bottle should burst.
] Heat the water bath to boiling, maintain at this temperature for 30 minutes, and then cool gradually to room temperature before handling the bottle. Remove the stopper, add phenolphthalein TS, and titrate the excess alkali with 1 N hydrochloric acid VS. Perform a blank determination (see Residual Titrations  541
541 ). Each mL of 1 N alcoholic potassium hydroxide is equivalent to 64.51 mg of C2H5Cl.
). Each mL of 1 N alcoholic potassium hydroxide is equivalent to 64.51 mg of C2H5Cl.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
USP32–NF27 Page 2330
Pharmacopeial Forum: Volume No. 31(5) Page 1368