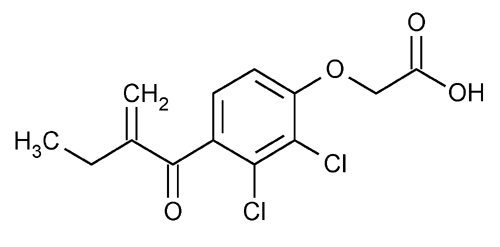
Ethacrynic Acid
Acetic acid, [2,3-dichloro-4-(2-methylene-1-oxobutyl)phenoxy]-.
[2,3-Dichloro-4-(2-methylenebutyryl)phenoxy]acetic acid
» Ethacrynic Acid contains not less than 97.0 percent and not more than 102.0 percent of C13H12Cl2O4, calculated on the dried basis.
Caution—Use care in handling Ethacrynic Acid, since it irritates the skin, eyes, and mucous membranes.
Packaging and storage—
Preserve in well-closed containers. Store at 25 , excursions permitted between 15
, excursions permitted between 15 and 30
and 30 .
.
Identification—
B: Ultraviolet Absorption  197U
197U —
—
Solution:
50 µg per mL.
Medium:
methanol.
Absorptivities at 271 nm, calculated on the dried basis, do not differ by more than 3.0%.
C:
Add 2 mL of 1 N sodium hydroxide to about 25 mg of it, and heat for several minutes in a boiling water bath. Cool the solution, acidify with 0.25 mL of 18 N sulfuric acid, add 0.5 mL of chromotropic acid sodium salt solution (1 in 10), then add, cautiously, 2 mL of sulfuric acid TS: a deep violet color is produced.
Loss on drying  731
731 —
Dry it at a pressure not exceeding 5 mm of mercury at 60
—
Dry it at a pressure not exceeding 5 mm of mercury at 60 for 2 hours: it loses not more than 0.25% of its weight.
for 2 hours: it loses not more than 0.25% of its weight.
Residue on ignition  281
281 :
not more than 0.1%.
:
not more than 0.1%.
Toluene extractives—
Accurately weigh about 1 g into a glass-stoppered, 100-mL cylinder. Add 50 mL of sodium sulfite solution (2 in 25), and agitate until the solid dissolves. Allow to stand for 20 minutes, add 5 mL of hydrochloric acid, and mix. Divide the solution between two centrifuge tubes, each of which contains 15 mL of toluene. Close each tube tightly, using a polyethylene stopper, and shake vigorously during 2 minutes, occasionally relieving the pressure from the sulfur dioxide by loosening the stoppers. Centrifuge the tubes, withdraw most of the upper layer by means of a syringe, avoiding withdrawal of any of the lower, aqueous phase, and transfer the toluene extracts to a tared evaporating dish. Repeat the extraction twice with additional 15-mL portions of toluene, and evaporate the combined extracts on a steam bath to dryness. Dry the residue at a pressure not exceeding 5 mm of mercury at 60 for 2 hours. Cool, and weigh: not more than 2.0% of extractives is found.
for 2 hours. Cool, and weigh: not more than 2.0% of extractives is found.
Equivalent weight—
Dissolve about 400 mg, accurately weighed, in 100 mL of methanol, add 5 mL of water, and titrate with 0.1 N sodium hydroxide VS, determining the endpoint potentiometrically, using a calomel–glass electrode system. Perform a blank determination, and make any necessary correction. Calculate the equivalent weight on the dried basis: it is between 294 and 309.
Heavy metals, Method II  231
231 :
0.001%.
:
0.001%.
Assay—
Dissolve about 100 mg of Ethacrynic Acid, accurately weighed, in 20 mL of glacial acetic acid in an iodine flask. Pipet 20 mL of 0.1 N bromine VS into the flask, add 3 mL of hydrochloric acid, immediately insert the stopper, and seal the flask with a few mL of water in the stopper well. Swirl the flask, and allow to stand in the dark for 1 hour. Add 50 mL of water and 15 mL of potassium iodide TS, and immediately titrate with 0.1 N sodium thiosulfate VS, adding 2.0 mL of starch TS as the endpoint is approached. Perform a blank determination (see Residual Titrations under Titrimetry  541
541 ). Each mL of 0.1 N bromine is equivalent to 15.16 mg of C13H12Cl2O4.
). Each mL of 0.1 N bromine is equivalent to 15.16 mg of C13H12Cl2O4.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Sujatha Ramakrishna, Ph.D.
Scientist 1-301-816-8349 |
(MDCV05) Monograph Development-Cardiovascular |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2318
Pharmacopeial Forum: Volume No. 29(5) Page 1479
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.