Esterified Estrogens
» Esterified Estrogens is a mixture of the sodium salts of the sulfate esters of the estrogenic substances, principally estrone. It is a dispersion of the estrogenic substances on a suitable powdered diluent. The content of total esterified estrogens is not less than 90.0 percent and not more than 110.0 percent of the labeled amount.
Esterified Estrogens contains not less than 75.0 percent and not more than 85.0 percent of sodium estrone sulfate, and not less than 6.0 percent and not more than 15.0 percent of sodium equilin sulfate, in such proportion that the total of these two components is not less than 90.0 percent, of the labeled amount of esterified estrogens.
Packaging and storage—
Preserve in tight containers.
Labeling—
Label it to state the content of Esterified Estrogens on a weight-to-weight basis.
Free steroids—
Proceed with Esterified Estrogens as directed in the test for Limit of estrone, equilin, and 17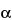 -dihydroequilin (free steroids) under Conjugated Estrogens. The limit is 3.0% of free steroids.
-dihydroequilin (free steroids) under Conjugated Estrogens. The limit is 3.0% of free steroids.
Assay—
Internal standard solution, Stock solution, Acetate buffer, pH 5.2, System suitability solution, Standard preparation, and Chromatographic system—
Proceed as directed in the Assay under Conjugated Estrogens.
Assay preparation—
Using an accurately weighed quantity of Esterified Estrogens, equivalent to about 2 mg of total esterified estrogens, proceed as directed for Assay preparation in the Assay under Conjugated Estrogens.
Procedure—
Proceed as directed in the Assay under Conjugated Estrogens. Calculate the quantity, in mg, of each sodium estrogen sulfate (estrone and equilin) in the portion of Esterified Estrogens taken by the formula:
(0.005)(F)Ci(RU / RS)
in which F is the factor converting free estrogen to the conjugate sodium salt, the factor being 1.377 for estrone and 1.380 for equilin; Ci is the concentration, in µg per mL, of USP Estrone RS or USP Equilin RS, as appropriate, in the alcohol solution; and RU and RS are the ratios of the estrone or equilin peak areas to the 3-O-methylestrone peak areas obtained from the Assay preparation and the Standard preparation, respectively.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Daniel K. Bempong, Ph.D.
Senior Scientist 1-301-816-8143 |
(MDPS05) Monograph Development-Pulmonary and Steroids |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 2312
Pharmacopeial Forum: Volume No. 32(6) Page 1678
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.