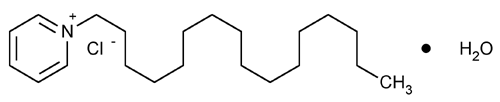
Cetylpyridinium Chloride
Pyridinium, 1-hexadecyl-, chloride, monohydrate.
1-Hexadecylpyridinium chloride monohydrate
Anhydrous 339.99
» Cetylpyridinium Chloride contains not less than 99.0 percent and not more than 102.0 percent of C21H38ClN, calculated on the anhydrous basis.
Packaging and storage—
Preserve in well-closed containers.
Identification—
Solution:
40 µg per mL.
Medium:
water.
C:
Dissolve 100 mg in 50 mL of water: a 10-mL portion of the solution responds to the tests for Chloride  191
191 , except that a turbidity is produced, rather than a curdy white precipitate, when the silver nitrate TS is added.
, except that a turbidity is produced, rather than a curdy white precipitate, when the silver nitrate TS is added.
Acidity—
Dissolve 500 mg, accurately weighed, in 50 mL of water, add phenolphthalein TS, and titrate with 0.020 N sodium hydroxide: not more than 2.5 mL is required for neutralization.
Water, Method I  921
921 :
between 4.5% and 5.5%.
:
between 4.5% and 5.5%.
Residue on ignition  281
281 :
not more than 0.2%, calculated on the anhydrous basis.
:
not more than 0.2%, calculated on the anhydrous basis.
Heavy metals, Method II  231
231 :
0.002%.
:
0.002%.
Pyridine—
Dissolve 1 g in 10 mL of sodium hydroxide solution (1 in 10) without heating: the odor of pyridine is not immediately perceptible.
Assay—
Transfer about 200 mg of Cetylpyridinium Chloride, accurately weighed, to a glass-stoppered, 250-mL graduated cylinder containing 75 mL of water. Add 10 mL of chloroform, 0.4 mL of bromophenol blue solution (1 in 2000), and 5 mL of a freshly prepared solution of sodium bicarbonate (4.2 in 1000), and titrate with 0.02 M sodium tetraphenylboron VS until the blue color disappears from the chloroform layer. Add the last portions of the sodium tetraphenylboron solution dropwise, agitating vigorously after each addition. Each mL of 0.02 M sodium tetraphenylboron is equivalent to 6.800 mg of C21H38ClN.
Auxiliary Information—
Please check for your question in the FAQs before contacting USP.
Chromatographic Column—
| Topic/Question | Contact | Expert Committee |
| Monograph | Behnam Davani, Ph.D., M.B.A.
Senior Scientist 1-301-816-8394 |
(MDAA05) Monograph Development-Antivirals and Antimicrobials |
| Reference Standards | Lili Wang, Technical Services Scientist 1-301-816-8129 RSTech@usp.org |
USP32–NF27 Page 1881
Chromatographic columns text is not derived from, and not part of, USP 32 or NF 27.