- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Adrenaline / Epinephrine |
 |
(Ph Eur monograph 2303)
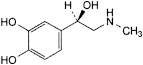
C9H13NO3 183.2 51-43-4
Adrenoceptor agonist.
Adrenaline Eye Drops/Epinephrine Eye Drops
Dilute Adrenaline Injection (1 in 10,000)/Dilute Epinephrine Injection (1 in 10,000)
Ph Eur
4-[(1R)-1-Hydroxy-2-(methylamino)ethyl]benzene-1,2-diol.
Synthetic product.
99.0 per cent to 101.0 per cent (dried substance).
White or almost white crystalline powder, becoming coloured on exposure to air and light.
Practically insoluble in water, in ethanol (96 per cent) and in methylene chloride. It dissolves in hydrochloric acid.
A. Infrared absorption spectrophotometry (2.2.24).
Comparison adrenaline CRS.
B. Specific optical rotation (see Tests).
Dissolve 1.000 g in a 25.75 g/L solution of hydrochloric acid R and dilute to 50.0 mL with the same solvent. Examine the solution immediately.
Solution S is not more opalescent than reference suspension II (2.2.1) and not more intensely coloured than reference solution BY5 (2.2.2, Method II).
- 50.0 to - 54.0 (dried substance), determined on solution S.
Liquid chromatography (2.2.29). Prepare the solutions protected from light.
Solvent mixture A Dissolve 5.0 g of potassium dihydrogen phosphate R and 2.6 g of sodium octanesulfonate R in water for chromatography R and dilute to 1000 mL with the same solvent (it is usually necessary to stir for at least 30 min to achieve complete dissolution). Adjust to pH 2.8 with phosphoric acid R.
Solvent mixture B acetonitrile R1, solvent mixture A (13:87 V/V).
Test solution Dissolve 40 mg of the substance to be examined in 5 mL of 0.1 M hydrochloric acid and dilute to 50.0 mL with solvent mixture B.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with solvent mixture B. Dilute 1.0 mL of this solution to 10.0 mL with solvent mixture B.
Reference solution (b) Dissolve 1.5 mg of noradrenaline tartrate CRS (impurity B) and 1.5 mg of adrenalone hydrochloride R (impurity C) in solvent mixture B, add 1.0 mL of the test solution and dilute to 100 mL with solvent mixture B.
Reference solution (c) Dissolve the contents of a vial of adrenaline impurity mixture CRS (containing impurities D and E) in 1.0 mL of the blank solution.
Reference solution (d) Dissolve 4 mg of adrenaline with impurity F CRS in 0.5 mL of 0.1 M hydrochloric acid and dilute to 5 mL with solvent mixture B.
Blank solution 0.1 M hydrochloric acid, solvent mixture B (1:9 V/V).
- — size: l = 0.10 m, Ø = 4.6 mm;
- — stationary phase: end-capped octadecylsilyl silica gel for chromatography R (3 µm);
- — temperature: 50 °C.
- — mobile phase A: acetonitrile R1, solvent mixture A (5:95 V/V);
- — mobile phase B: acetonitrile R1, solvent mixture A (45:55 V/V);
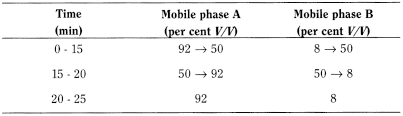
Flow rate 2.0 mL/min.
Detection Spectrophotometer at 210 nm.
Injection 20 µL.
Identification of impurities Use the chromatogram supplied with adrenaline impurity mixture CRS and the chromatogram obtained with reference solution (c) to identify the peaks due to impurities D and E; use the chromatogram supplied with adrenaline with impurity F CRS and the chromatogram obtained with reference solution (d) to identify the peak due to impurity F.
Relative retention With reference to adrenaline (retention time = about 4 min): impurity F = about 0.2; impurity B = about 0.8; impurity C = about 1.3; impurity D = about 3.3; impurity E = about 3.7.
System suitability Reference solution (b):
- — resolution: minimum 3.0 between the peaks due to impurity B and adrenaline.
- — correction factors: for the calculation of content, multiply the peak areas of the following impurities by the corresponding correction factor: impurity D = 0.7; impurity E = 0.6;
- — impurities B, C, F: for each impurity, not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent);
- — impurities D, E: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent);
- — unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent);
- — total: not more than 5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent);
- — disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Maximum 0.5 per cent, determined on 1.000 g by drying over diphosphorus pentoxide R at a pressure not exceeding 0.7 kPa for 18 h.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.150 g in 50 mL of anhydrous acetic acid R. Titrate with 0.1 M perchloric acid, determining the end-point potentiometrically (2.2.20).
1 mL of 0.1 M perchloric acid is equivalent to 18.32 mg of C9H13NO3.
Under nitrogen, protected from light.
Specified impurities B, C, D, E, F.
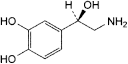
B. (1R)-2-amino-1-(3,4-dihydroxyphenyl)ethanol (noradrenaline),
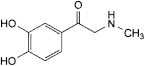
C. 1-(3,4-dihydroxyphenyl)-2-(methylamino)ethanone (adrenalone),
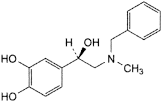
D. 4-[(1R)-2-(benzylmethylamino)-1-hydroxyethyl]benzene-1,2-diol,
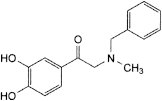
E. 2-(benzylmethylamino)-1-(3,4-dihydroxyphenyl)ethanone,
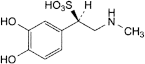
F. (1R)-1-(3,4-dihydroxyphenyl)-2-(methylamino)ethanesulfonic acid.
_ Ph Eur

