- British Pharmacopoeia Volume I & II
- Monographs: Medicinal and Pharmaceutical Substances
Ambroxol Hydrochloride |
 |
(Ph. Eur. monograph 1489)
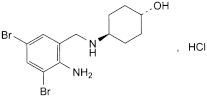
C13H18Br2N2O,HCl 414.6 23828-92-4
Mucolytic expectorant.
Ph Eur
trans-4-[(2-Amino-3,5-dibromobenzyl)amino]cyclohexanol hydrochloride.
99.0 per cent to 101.0 per cent (dried substance).
White or yellowish, crystalline powder.
Sparingly soluble in water, soluble in methanol, practically insoluble in methylene chloride.
First identification B, D.
Second identification A, C, D.
A. Ultraviolet and visible absorption spectrophotometry (2.2.25).
Test solution Dissolve 20.0 mg in 0.05 M sulfuric acid and dilute to 100.0 mL with the same acid. Dilute 2.0 mL of the solution to 10.0 mL with 0.05 M sulfuric acid.
Spectral range 200-350 nm.
Absorption maxima At 245 nm and 310 nm.
Absorbance ratio A245/A310 = 3.2 to 3.4.
B. Infrared absorption spectrophotometry (2.2.24).
Comparison ambroxol hydrochloride CRS.
C. Thin-layer chromatography (2.2.27).
Test solution Dissolve 50 mg of the substance to be examined in methanol R and dilute to 5 mL with the same solvent.
Reference solution Dissolve 50 mg of ambroxol hydrochloride CRS in methanol R and dilute to 5 mL with the same solvent.
Plate TLC silica gel F254 plate R.
Mobile phase concentrated ammonia R, propanol R, ethyl acetate R, hexane R (1:10:20:70 V/V/V/V).
Application 10 µL.
Development Over 2/3 of the plate.
Drying In air.
Detection Examine in ultraviolet light at 254 nm.
Results The principal spot in the chromatogram obtained with the test solution is similar in position and size to the principal spot in the chromatogram obtained with the reference solution.
D. Dissolve 25 mg in 2.5 mL of water R, mix with 1.0 mL of dilute ammonia R1 and allow to stand for 5 min. Filter and acidify the filtrate with dilute nitric acid R. The filtrate gives reaction (a) of chlorides (2.3.1).
Dissolve 0.75 g in methanol R and dilute to 15 mL with the same solvent.
Solution S is clear (2.2.1) and not more intensely coloured than reference solution Y6 (2.2.2, Method II).
4.5 to 6.0.
Dissolve 0.2 g in carbon dioxide-free water R and dilute to 20 mL with the same solvent.
Liquid chromatography (2.2.29). Prepare the solutions immediately before use.
Test solution Dissolve 50 mg of the substance to be examined in water R and dilute to 50.0 mL with the same solvent.
Reference solution (a) Dilute 1.0 mL of the test solution to 100.0 mL with water R. Dilute 1.0 mL of this solution to 10.0 mL with the mobile phase.
Reference solution (b) In order to prepare impurity B in situ, dissolve 5 mg of the substance to be examined in 0.2 mL of methanol R, add 0.04 mL of a mixture of 1 volume of formaldehyde solution R and 99 volumes of water R. Heat at 60 °C for 5 min. Evaporate to dryness under a current of nitrogen. Dissolve the residue in 5 mL of water R and dilute to 20.0 mL with the mobile phase.
- — size: l = 0.25 m, Ø = 4.0 mm;
- — stationary phase: octadecylsilyl silica gel for chromatography R (5 µm).
Mobile phase A mixture of equal volumes of acetonitrile R and a solution prepared as follows: dissolve 1.32 g of ammonium phosphate R in 900 mL of water R, adjust to pH 7.0 with phosphoric acid R and dilute to 1000 mL with water R.
Flow rate 1 mL/min.
Detection Spectrophotometer at 248 nm.
Injection 20 µL.
Run time 3 times the retention time of ambroxol.
Identification of impurities Use the chromatogram obtained with reference solution (b) to identify the peak due to impurity B.
Relative retention With reference to ambroxol (retention time = about 9 min): impurity B = about 0.6.
System suitability Reference solution (b):
- — resolution: minimum 4.0 between the peaks due to impurity B and ambroxol.
- — unspecified impurities: for each impurity, not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.10 per cent),
- — total: not more than 3 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.3 per cent),
- — disregard limit: 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent).
Maximum 20 ppm.
1.0 g complies with test C. Prepare the reference solution using 2 mL of lead standard solution (10 ppm Pb) R.
Maximum 0.5 per cent, determined on 1.000 g by drying in an oven at 105 °C.
Maximum 0.1 per cent, determined on 1.0 g.
Dissolve 0.300 g in 70 mL of ethanol (96 per cent) R and add 5 mL of 0.01 M hydrochloric acid. Carry out a potentiometric titration (2.2.20), using 0.1 M sodium hydroxide. Read the volume added between the 2 points of inflexion.
1 mL of 0.1 M sodium hydroxide is equivalent to 41.46 mg of C13H19Br2ClN2O.
Protected from light.
Other detectable impurities (the following substances would, if present at a sufficient level, be detected by one or other of the tests in the monograph. They are limited by the general acceptance criterion for other/unspecified impurities and/or by the general monograph Substances for pharmaceutical use (2034). It is therefore not necessary to identify these impurities for demonstration of compliance. See also 5.10. Control of impurities in substances for pharmaceutical use): A, B, C, D, E.
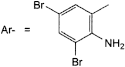
A. Ar-CH2OH: (2-amino-3,5-dibromophenyl)methanol,
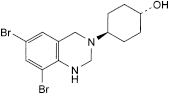
B. trans-4-(6,8-dibromo-1,4-dihydroquinazolin-3(2H)-yl)cyclohexanol,
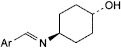
C. trans-4-[[(E)-2-amino-3,5-dibromobenzyliden]amino]cyclohexanol,
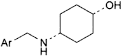
D. cis-4-[(2-amino-3,5-dibromobenzyl)amino]cyclohexanol,
E. Ar-CH=O: 2-amino-3,5-dibromobenzaldehyde.
Ph Eur

